| Indomethacin Sorbitol Ester 1:Primoryl Hydroxyl |
Product Code: I042016
English Name: Indomethacin Sorbitol Ester 1:Primoryl Hydroxyl
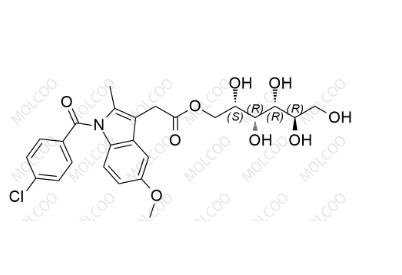
English Alias: (2S,3R,4R,5R)-2,3,4,5,6-pentahydroxyhexyl 2-(1-(4-chlorobenzoyl)-5-methoxy-2-methyl-1H-indol-3-yl)acetate
CAS No.: Not provided
Molecular Formula: C₂₅H₂₈ClNO₉
Molecular Weight: 521.94
As an ester derivative of indomethacin and sorbitol, it combines the anti-inflammatory activity of indomethacin with the hydrophilicity of sorbitol, potentially improving the water solubility and bioavailability of the parent drug.
The structure contains polyhydroxyl functional groups, which can be used as a novel prodrug or excipient for formulation development, providing more possibilities for drug delivery systems.
Pharmaceutical Formulation R&D: Used to develop sustained-release or targeted formulations of indomethacin, regulating drug release rate through esterification modification to reduce gastrointestinal irritation.
Prodrug Research: As a prodrug candidate, study its hydrolytic metabolic pathway and activity conversion in vivo to optimize pharmacokinetic properties.
Excipient Compatibility Study: Evaluate its compatibility with other drug components as a functional excipient for (compound formulation) development.
Indomethacin is a non-steroidal anti-inflammatory drug (NSAID) commonly used in treating inflammatory diseases such as arthritis, but it suffers from poor water solubility and significant gastrointestinal side effects. Esterification with sorbitol to form derivatives aims to improve its physicochemical properties (e.g., solubility, stability) while retaining or enhancing efficacy. The research background of such derivatives is closely related to the clinical pain points of NSAID drugs, i.e., improving therapeutic compliance and safety through molecular modification.
Synthesis Process: Currently mainly prepared by esterification reaction, with research focusing on optimizing reaction conditions (such as catalyst, temperature) to improve yield and purity.
Efficacy Research: In vitro experiments show that some derivatives have anti-inflammatory activity comparable to indomethacin, but their in vivo metabolic mechanisms (such as ester bond hydrolysis rate) still need further verification.
Formulation Application: Preliminary studies indicate that such esters can achieve controlled release through solid dispersion or microsphere formulations, and related dosage forms are in the preclinical evaluation stage.
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!