-
外観
無色~うすい黄色, 澄明の液体
-
溶解性
水に易溶。有機溶剤に可溶, 但し反応性が強い。
-
解説
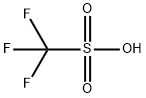
トリフルオロメタンスルホン酸,トリフルオロメタンスルホンサン,CHF3O3S(150.07).CF3SO3H.略称TFSA.メタンスルホニルクロリドの電解フッ素化によって得られるトリフルオロメタンスルホニルフルオリドの加水分解で合成される.無色の液体.融点-34 ℃,沸点162 ℃.d251.698.空気中で白煙を生じ,一水和物結晶になる.水,有機溶媒に可溶.硫酸より強い超強酸の一種で,有機合成反応触媒として用いられる.皮膚や眼をはげしく刺激する.
-
用途
試薬,触媒,医薬,農薬原料
-
用途
有機合成原料(重合触媒、スルホン化剤、脱水剤)、溶媒(たん白質、ポリマー)。
-
化学的特性
Clear very light yellow liquid. Smoke in the air, easy to absorb water to form a hydrate. It is easily soluble in water, releases a lot of heat, and hydrolyzes to generate trifluoromethane (CHF3) and sulfuric acid.
-
使用
Trifluoromethanesulfonic acid acts as a catalyst for esterification reactions and an acidic titrant in nonaqueous acid-base titration. It is useful in protonations due to the presence of conjugate base triflate is non nucleophilic. It serves as a deglycosylation agent for glycoproteins. In addition, it is a precursor and a catalyst in organic chemistry. It reacts with acyl halides to prepare mixed triflate anhydrides, which are strong acylating agents used in Friedel-Crafts reactions. It acts as a key starting material for the preparation of ethers and olefins by reacting with alcohols as well as to prepare trifluoromethanesulfonic anhydride by dehydration reaction.
-
定義
ChEBI: Trifluoromethanesulfonic acid is a one-carbon compound that is methanesulfonic acid in which the hydrogens attached to the methyl carbon have been replaced by fluorines. It is a one-carbon compound and a perfluoroalkanesulfonic acid. It is a conjugate acid of a triflate.
-
一般的な説明
Trifluoromethanesulfonic acid is a strong organic acid. It can be prepared by reacting bis(trifluoromethylthio)mercury with H2O2. On mixing with HNO3, it affords a nitrating reagent (a nitronium salt). This reagent is useful for the nitration of aromatic compounds. Its dissociation in various organic solvents has been studied.
-
安全性プロファイル
A corrosive irritant to
the skin, eyes, and mucous membranes. A
strong acid. Violent reaction with acyl
chlorides or aromatic hydrocarbons evolves
toxic hydrogen chloride gas. When heated to
decomposition it emits toxic fumes of Fand
SOx. See also FLUORIDES.