Amyloride is also a potassium sparing diuretic that exhibits moderate activity. It is not an
antagonist of aldosterone. It inhibits reabsorption of sodium ions and reduces excretion of potassium ions.
Amiloride is an epithelial sodium channel blocker.
Amyloride is rarely used individually—as a rule it is used in combination
with thiazides or loop diuretics. It is mainly used in combination with thiazide diuretics
for cardiac insufficiency and hypertension, especially in cases where it is necessary to
prevent hypokalemia.
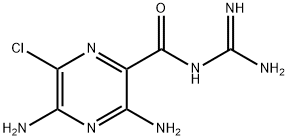
ChEBI: A monocarboxylic acid amide that is N-carbamimidoylpyrazine-2-carboxamide substituted by amino groups at positions 3 and 5 and a chloro group at position 6.
Step A: Preparation of methyl 3-amino-5,6-dichloropyrazinoare: Methyl 3-
aminopyrazinoate (765 g, 5 mols) is suspended in 5 liters of dry benzene.
While stirring under anhydrous conditions sulfuryl chloride (1.99 liters, 3.318
g, 24.58 mols) is added over a period of 30 minutes and stirring is continued
for 1 hour. During this period, the temperature rises to about 50°C and then
begins to drop. The mixture is heated cautiously to reflux (60°C), refluxed for
5 hours and then stirred overnight at room temperature. The excess sulfuryl
chloride is distilled off at atmospheric pressure (distillation is stopped when
vapor temperature reaches 78%). The dark red mixture is chilled to 6°C. The
crystals are filtered off, washed by displacement with two 100 ml portions of
cold (8°C) benzene, then washed with 300 ml petroleum ether and dried in
vacuum at room temperature, yielding 888 g (80%) of methyl 3-amino-5,6-
dichloropyrazinoate in the form of red crystals, MP 228-230°C. The crude
product is dissolved in 56 liters of boiling acetonitrile and passed through a
heated (70-80°C) column of decolorizing charcoal (444 g). The column is
washed with 25 liters of hot acetonitrile, the combined eluate concentrated in
vacuum to about 6 liters and chilled to 5°C. The crystals that form are
filtered, washed three times with cold acetonitrile, and air dried to constant
weight, yielding 724 g (82% recovery, 66% overall) of methyl 3-amino-5,6-
dichloropyrazinoate in the form of yellow crystals, MP 230-234°C. After
additional recrystallizations from acetonitrile the product melts at 233-234°C.
Step B: Preparation of methyl-3,5diamino-6-chloropyrazinoete: In a 2-liter, 3-necked flask fitted with a a mechanical stirrer, thermometer and gas inlet tube
is placed dry dimethyl sulfoxide (1 liter). Methyl 3-amino-5,6-
dichloropyrazinoate (100 g, 0.45 mol) is added and the mixture stirred and
heated at 65°C on a steam bath until solution is effected. A stream of dry
ammonia gas is admitted to the solution with continuous stirring, over a
period of 45 minutes while the temperature is maintained at 65-70°C. The
solution is cooled to about 10°C with continuous stirring and ammonia gas is
admitted for an additional 1 1/4 hours. The yellow reaction mixture is poured,
with stirring, into cold water (2 liters) and the light yellow solid that separates
is removed by filtration, thoroughly washed with water, and dried in a vacuum
desiccator to give 82.5 g (91%) of methyl 3,5-diamino-6-chloropyrazinoate,
MP 210-212°C. Recrystallization from acetonitrile gives material melting at
212-213°C.
Step C: Preparation of the base: A 300 ml one-necked, round-bottomed flask,
equipped with a water-cooled condenser, calcium chloride tube and magnetic
stirrer is charged with anhydrous methanol (150 ml) and sodium metal (5.75
g, 0.25 g atom). When the reaction is complete, the solution is treated with
dry guanidine hydrochloride (26.3 g, 0.275 mol) and stirred for 10 minutes.
The sodium chloride that forms is removed by filtration. The solution is
concentrated in vacuum to a volume of 30 ml and the residue treated with the
product of Step B, heated one minute on a steam bath and kept at 25°C for 1
hour. The product is filtered, washed well with water, dissolved in dilute
hydrochloric acid and the free base precipitated by addition of sodium
hydroxide to give the amiloride product base, a solid which melts at 240.5-
241.5°C.
To produce the hydrochloride, the base is suspended in water (70 ml) and
treated with sufficient 6 N hydrochloric acid to dissolve the free base. The
solution is filtered and treated with concentrated hydrochloric acid (5 ml). The
hydrochloride salt (22 g, 97%) separates and is recrystallized from water (50
ml) containing concentrated hydrochloric acid (3 ml).
Amiloride, another potassium-sparing diuretic, is an aminopyrazine structurally related to triamterene as an open-chain analogue. Similar to triamterene, it interferes with
the process of cationic exchange in the distal convoluted tubule by blocking luminal sodium channels. It blocks the reabsorption of sodium ion and the secretion of
potassium ion. It has no effect on the action of aldosterone. Oral amiloride is approximately 50% absorbed, with
a duration of action of 10 to 12 hours, which is slightly longer than that for triamterene. Although triamterene is extensively metabolized, approximately 50% of amiloride
is excreted unchanged. Renal impairment can increase its elimination half-life. Like triamterene, amiloride combined with a thiazide or loop diuretic is used to treat edema
or hypertension.
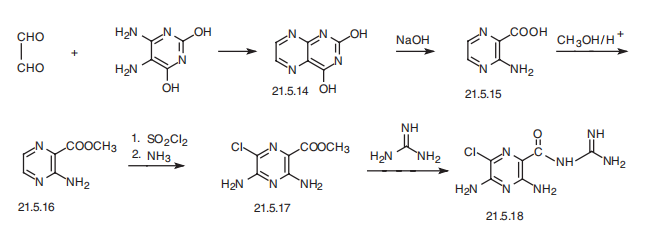
Amyloride, N-amidino-3,5-diamino-6-chloropirazincarboxamide (21.5.18), is
synthesized from 5,6-diaminouracil, which upon reaction with glyoxal transforms into a
pyrazineopyrimidine derivative (21.5.14), which decomposes upon further reaction with a
strong alkaline, forming 3-aminopirazin-2-carboxylic acid (21.5.15). This is esterified into
the corresponding methyl ester (21.5.16), and subsequently treated with sulfonyl chloride
and ammonia, which gives the methyl ester of 3,5-diamino-6-chloropirazin-2-carboxylic
acid (21.5.17). Reacting this with guanidine gives amyloride (21.5.18).
This potassium-sparing diuretic (FW = 229.63 g/mol; CAS 2016-88-8;
Absorbance at 10 mg/mL (water) = 642 at 212 nm, 555 at 285 nm, and 617
at 362 nm; pKa = 8.7; Soluble in hot water (50 mg/mL), yielding a clear,
yellow-green solution), also known as MK 870, Midamor, and 3,5-
diamino-6-chloro-N- (diaminomethylene) pyrazine-2-carboxamide, is an
epithelial sodium channel (or ENaC) blocker used to manage hypertension
and congestive heart failure (1-4). Two classes of Na+ transporters are
sensitive to this drug: (a) the conductive Na+ entry pathway found in
electrically high resistance epithelia and (b) a Na+-H+ electroneutral
exchange system found in certain leaky epithelia, such as the renal
proximal tubule. Amiloride exhibits a Ki value of <1 μM for the kidney
transporter and approximately 1 mM in the colon. Midamor is a potassium-
conserving (antikaliuretic) drug that possesses weak (compared with
thiazide diuretics) natriuretic, diuretic, and antihypertensive activity. Like
other potassium-conserving agents, amiloride may cause hyperkalemia
(i.e., serum K+ levels >5.5 mEq/L), which, if uncorrected, can be fatal.
Amiloride-induced hyperkalemia occurs in ~10% of patients, when used
without a kaliuretic diuretic. This complication is more frequent in patients
showing evidence of renal impairment and diabetes mellitus. When used
along with a thiazide diuretic in patients without these complications, the
risk of Amiloride-induced hyperkalemia drops to 1-2%. Amiloride is not
metabolized by the liver and is instead excreted unchanged by the kidney.
Amiloride displaces both adenosine A1 receptor agonist and antagonist
binding with a Ki value in the low micromolar range, when assayed in calf
brain membranes. Inhibition is counteracted by NaCl and protons.
Amiloride (IC50 = 0.25 mM) and 5- (N-ethyl-N-isopropyl) amiloride (IC50 =
0.11 mM) also inhibit coxsackievirus B3 RNA polymerase in a single-
nucleotide incorporation assay, although the argument that amiloride
competes with incoming nucleoside triphosphates is unconvincing. A high-
fidelity RNA dependent CVB3 RNA polymerase (RdRp) variant is
amiloride-resistant.