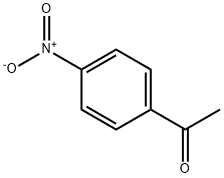
The pure product is pale yellow crystal or needle crystal. Soluble in hot ethanol, ether and benzene, insoluble in water. 4-Nitroacetophenone is the raw material for the manufacture of synthomycin and chloramphenicol.
4''-Nitroacetophenone is used as a reagent in the synthesis of 4-Nitroacetophenone thiosemicarbazone derivatives and their copper(II) complexes which have potential anti-trypanosomal activity in vitro. Also used as a reagent in the synthesis of (R)-(4-Nitrophenyl)oxirane (N504430) and (S)-(4-Nitrophenyl)oxirane (N504435).
ChEBI: 4-nitroacetophenone is a member of the class of acetophenones that is acetophenone substituted at the para-position by a nitro group. It is a C-nitro compound and a member of acetophenones.
In the presence of catalyst cobalt stearate, p-nitroethylbenzene is oxidized to synthesize 4-nitroacetophenone with air at 140-150°C and 0.2MPa pressure. The reaction product is washed with water, neutralized, centrifugally dehydrated, and dried to obtain a finished product.
One of several alternative routes to syn�thetic chloroamphenicol uses 4-nitroacetophenone as start�ing material with a first-stage bromination to α-bromo-4-nitroacetophenone followed by buildup of the side chain. α-bromo-4-nitroacetophenone is the key precursor in the synthesis of the bronchodilator clenbuterol.
P-nitroacetophenone appears as yellow prisms or bright yellow powder. (NTP, 1992)
A nitrated ketone. Ketones are reactive with many acids and bases liberating heat and flammable gases (e.g., H2). The amount of heat may be sufficient to start a fire in the unreacted portion of the ketone. Ketones react with reducing agents such as hydrides, alkali metals, and nitrides to produce flammable gas (H2) and heat. Ketones are incompatible with isocyanates, aldehydes, cyanides, peroxides, and anhydrides. They react violently with aldehydes, HNO3, HNO3 + H2O2, and HClO4.
Flash point data for 4-Nitroacetophenone are not available. 4-Nitroacetophenone is probably combustible.
Crystallise the ketone from EtOH or aqueous EtOH. [Beilstein 7 IV 657.]