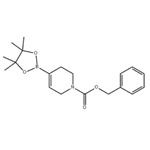
N-Benzyloxycarbonyl-3,6-dihydro-2H-pyridine-4-boronic acid pinacol ester was synthesized from bis(pinacolato) boronic acid and the compound (CAS: 286961-24-8) according to the method reported in Tetrahedron Lett. The steps were as follows: bis(pinacolato)ethylborane (338 mg, 1.33 mmol), potassium acetate (356 mg, 3.63 mmol), [1,1'-bis(diphenylphosphino)ferrocene]palladium(II) dichloride (PdCl2dppf; 30 mg, 0.04 mmol), and 1,1'-bis(diphenylphosphino)ferrocene ( 20 mg, 0.04 mmol), followed by addition of the product of Example 58A (440 mg, 1.21 mmol) in degassed 1,4-dioxane (7 mL). The reaction mixture was heated at 80 °C for 16 h, after which it was cooled to 23 °C, diluted with water and extracted with dichloromethane (3×). The organic phases were combined, dried over anhydrous sodium sulfate, filtered and the filtrate concentrated under reduced pressure. The residue was purified by fast column chromatography on silica gel (eluent: 20% ethyl acetate/hexane) to afford the target product N-benzyloxycarbonyl-3,6-dihydro-2H-pyridine-4-boronic acid pinacol ester (323 mg, 78% yield). The product was confirmed by 1H NMR (300 MHz, CDCl3) and mass spectrometry (ESI): 1H NMR δ 1.25 (s, 12H), 2.24 (m, 2H), 3.52 (dd, 2H, J = 5.7, 5.7 Hz), 4.03 (dd, 2H, J = 6 Hz), 5.14 (s, 2H), 6.46 (br m, 1H), 7.32 (m, 5H); MS (ESI) m/e 344 (M + H)+.
![4-(4,4,5,5-TETRAMETHYL-[1,3,2]DIOXABOROLAN-2-YL)-3,6-DIHYDRO-2H-PYRIDINE-1-CARBOXYLIC ACID BENZYL ESTER Structure](/CAS/GIF/286961-15-7.gif)

![1(2H)-Pyridinecarboxylic acid, 3,6-dihydro-4-[[(trifluoroMethyl)sulfonyl]oxy]-, phenylMethyl ester](/CAS/GIF/286961-24-8.gif)

![4-(4,4,5,5-TETRAMETHYL-[1,3,2]DIOXABOROLAN-2-YL)-3,6-DIHYDRO-2H-PYRIDINE-1-CARBOXYLIC ACID BENZYL ESTER pictures](/ProductImageEN/2021-04/Small/8c5a7b36-4b20-496b-965b-6b303650474b.jpg)