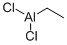
ETHYLALUMINUM DICHLORIDE
- Product NameETHYLALUMINUM DICHLORIDE
- CAS563-43-9
- MFC2H5AlCl2
- MW126.95
- EINECS209-248-6
- MOL File563-43-9.mol
Chemical Properties
| Melting point | 32 °C(lit.) |
| Boiling point | 68-70°C |
| Density | 0.927 g/mL at 25 °C |
| vapor pressure | 5 mm Hg ( 60 °C) |
| Flash point | 40 °F |
| storage temp. | 0-6°C |
| form | Liquid |
| color | Colorless to pale yellow |
| Specific Gravity | 0.927 |
| Water Solubility | Soluble in acetone. Miscible with alcohol, chloroform, ether. Reacts with water. |
| FreezingPoint | 32℃ |
| Sensitive | Air & Moisture Sensitive |
| Hydrolytic Sensitivity | 8: reacts rapidly with moisture, water, protic solvents |
| Merck | 326 |
| BRN | 4123357 |
| Exposure limits | ACGIH: TWA 50 ppm (Skin) OSHA: TWA 500 ppm(1800 mg/m3) NIOSH: IDLH 1100 ppm; TWA 50 ppm(180 mg/m3) |
| InChI | 1S/C2H5.Al.2ClH/c1-2;;;/h1H2,2H3;;2*1H/q;+2;;/p-2 |
| InChIKey | UAIZDWNSWGTKFZ-UHFFFAOYSA-L |
Safety Information
| Hazard Codes | F,T,N,C |
| Risk Statements | 11-15-23/24/25-34-14-67-65-62-51/53-48/20-14/15-17-63 |
| Safety Statements | 16-26-36/37/39-45-62-61-43-27 |
| RIDADR | UN 3399 4.3/PG 1 |
| WGK Germany | 1 |
| RTECS | BD0705000 |
| F | 10-21 |
| TSCA | TSCA listed |
| HazardClass | 4.3 |
| PackingGroup | I |
| HS Code | 29319090 |
| Storage Class | 4.3 - Hazardous materials which set free flammable gases upon contact with water |
| Hazard Classifications | Aquatic Chronic 3 Asp. Tox. 1 Eye Dam. 1 Flam. Liq. 2 Repr. 2 Skin Corr. 1B STOT RE 2 STOT SE 3 Water-react 1 |
| Hazardous Substances Data | 563-43-9(Hazardous Substances Data) |


