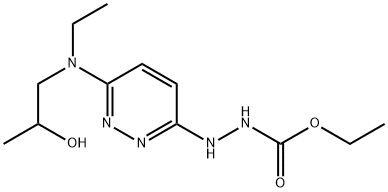
Crystallized in acetone, melting point 160-162°C. pKa 6.0. UV max absorption: 248, 340m (ε22100, 2250). Solubility (mg/m1): water 1.3, hydrochloric acid 235.0, dimethyl sulfoxide 323.0, methanol 21.0, dioxane 18.6, chloroform 8.5, ether, benzene, and cyclohexane <0.1. Acute Toxicity LD50 rat, dog (mg/kg): 269, about 400 IV; 2060, >2000 oral. Acute toxicity LD50 rat (mg/kg): 700 intraperitoneal.
World Health Organization (WHO)
Cadralazine, a peripheral vasodilator, was introduced in 1989 for
the treatment of arterial hypertension.In 1992, its association with serious side
effects led to the refusal of registration in Norway. Animal experiments have
demonstrated drug-related impairment of thyroid function as well as potential carcinogenicity and genotoxicity. It remains available for treatment of hypertension
in Italy.