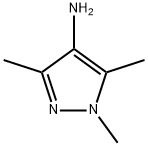
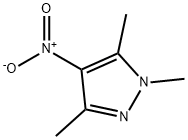
General procedure for the synthesis of 4-amino-1,3,5-trimethylpyrazole from 4-nitro-1,3,5-trimethylpyrazole: To a solution of 1,3,5-trimethyl-4-nitro-1H-pyrazole (2.1 g, 1.35 mmol) in ethanol (30 mL) was added powdered iron (14.85 g, 27 mmol) and 6N aqueous hydrochloric acid solution (0.23 mL. 0.67 mmol) and the mixture was refluxed for 16 hours. After completion of the reaction (monitored by TLC), the solvent was removed by evaporation under reduced pressure. The crude product was alkalized with 0.5 N aqueous sodium hydroxide and extracted with 10% methanol-dichloromethane (10%, 50 mL) solution. The organic layer was dried with anhydrous sodium sulfate, filtered and the solvent evaporated to afford 4-amino-1,3,5-trimethylpyrazole as a beige solid (1.68 g, 99% yield).' HNMR (400 MHz, DMSO-d6): δ 3.50 (s, 3H), 3.27 (bs, 2H), 2.03 (s, 3H), 1.94 (s, 3H).