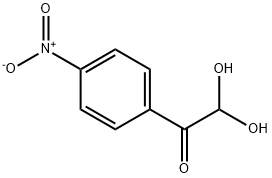
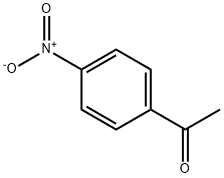
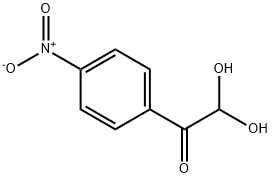
Synthesis
GENERAL METHODS: A mixture of 4-nitroacetophenone (0.6 mmol) and ethylene bromide (0.06 mmol) in DMSO (1.1 mL) was placed in an open-top flask and heated in an oil bath (see Table 2 for specific temperatures and reaction times). After completion of the reaction (monitored by TLC), the reaction mixture was diluted with water (11 mL), salted with saturated NaCl solution and extracted with ethyl acetate (5 × 4 mL). Only in case of synthesis of arylglyoxal, the organic layer was additionally washed with aqueous Na2CO3 (0.12 mmol, dissolved in 2 mL of water), followed by saturated aqueous NaCl solution and dried with anhydrous Na2SO4. The extract was purified by column chromatography (eluent: petroleum ether/ethyl acetate gradient) to give pure 2,2-dihydroxy-1-(4-nitrophenyl)ethanone.
References
[1] Tetrahedron Asymmetry, 2010, vol. 21, # 18, p. 2244 - 2248
[2] Tetrahedron, 2011, vol. 67, # 41, p. 8000 - 8008
[3] Journal of Fluorine Chemistry, 2010, vol. 131, # 12, p. 1289 - 1296
[4] Tetrahedron Letters, 2011, vol. 52, # 31, p. 4036 - 4038
[5] Tetrahedron, 2013, vol. 69, # 1, p. 22 - 28