Tobramycin
- Product NameTobramycin
- CAS32986-56-4
- CBNumberCB7154445
- MFC18H37N5O9
- MW467.52
- EINECS251-322-5
- MDL NumberMFCD00077885
- MOL File32986-56-4.mol
- MSDS FileSDS
Chemical Properties
| Melting point | 178 °C |
| alpha | D20 +129° (c = 1 in water) |
| Boiling point | 570.01°C (rough estimate) |
| Density | 1.3458 (rough estimate) |
| refractive index | 143 ° (C=4, H2O) |
| storage temp. | Keep in dark place,Inert atmosphere,2-8°C |
| solubility | H2O: 50 mg/mL, clear, faintly yellow |
| pka | pKa 6.7 (Uncertain);8.3 (Uncertain);9.9 (Uncertain) |
| form | White to off-white solid |
| color | white to off-white |
| Water Solubility | Soluble in water |
| Merck | 14,9490 |
| BRN | 1357507 |
| Stability | Hygroscopic |
| Major Application | pharmaceutical pharmaceutical small molecule |
| InChIKey | NLVFBUXFDBBNBW-TYINOFMDNA-N |
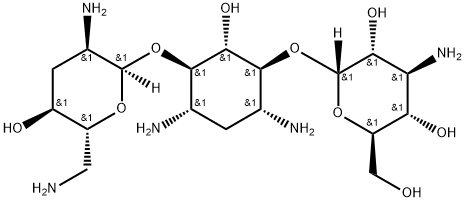
| SMILES | NC[C@H]1O[C@H](O[C@@H]2[C@@H](N)C[C@@H](N)[C@H](O[C@H]3O[C@H](CO)[C@@H](O)[C@@H](N)[C@H]3O)[C@H]2O)[C@H](N)C[C@@H]1O |
| FDA UNII | VZ8RRZ51VK |
Safety
| Symbol(GHS) |

|
|||||||||
| Signal word | Warning | |||||||||
| Hazard statements | H361f | |||||||||
| Precautionary statements | P201-P202-P280-P308+P313-P405-P501 | |||||||||
| Hazard Codes | Xi | |||||||||
| Risk Statements | 36/37/38 | |||||||||
| Safety Statements | 26-37/39 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | WK2100000 | |||||||||
| F | 3-10 | |||||||||
| HS Code | 29419090 | |||||||||
| Storage Class | 11 - Combustible Solids | |||||||||
| Hazard Classifications | Repr. 2 | |||||||||
| Hazardous Substances Data | 32986-56-4(Hazardous Substances Data) | |||||||||
| Toxicity | LD50 in mice, rats (mg/kg): 441, 969 s.c. (Welles) | |||||||||
| NFPA 704: |
|