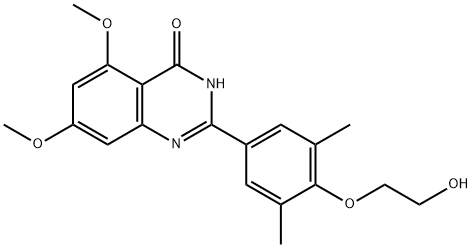
Description
The bromodomain and extra terminal (BET) proteins interact with acetylated lysine-containing sequences on target proteins
via their bromodomains, commonly altering gene transcription. The human BET proteins contain two bromodomains, the first (BD1) being closer to the N-terminus than the second (BD2). RVX-208 is a selective antagonist of BET bromodomains, binding with 10-100-fold higher affinity for BD2 (IC
50 = 0.04-0.28 μM) over BD1 (IC
50 = 1.8-3.1 μM). RVX-208 causes the selective release of BET proteins from chromatin in cells. It interferes with the BET protein BRD4, resulting in an increased expression of apolipoprotein (Apo) A
1 in cells, mice, monkeys, and humans. RVX-208 also reduces atherosclerosis in hyperlipidemic ApoE-deficient mice.
Uses
RVX-208 is a selective antagonist of bromodomain and extra terminal (BET)’s bromodomains.
in vivo
In the atherosclerosis prophylactic treatment study design, mice are fed a Western diet concurrent with the treatment with 150 mg/kg/dose b.i.d. for 12 weeks. Mice are sacrificed at 12 weeks after treatment. There is a progressive increase in body weight in both the vehicle treated as well as the Apabetalone (RVX-208) treated groups. However, there is only an increase of 4 g (from 24 g to 28 g) body weight after 12 weeks on Western diet in the Apabetalone treated group whereas this increase is found to be 9 g (25 g-34 g) in the vehicle treated group. The significant decrease in body weight gain in Apabetalone treated mice is not due to decreased feed consumption, suggesting a positive attribute of the molecule. Plasma lipid measurements are done at 6 weeks and 12 weeks of treatment with either the vehicle or Apabetalone. Compared to the vehicle control animals, Apabetalone treated mice show significant increase (~200%) in the levels of HDL-C at 6 weeks of treatment, which is sustained until end of the study (12 weeks)[3].
Background
RVX-208 is a potent and selective inhibitor of bromodomain and extra terminal proteins, with a much stronger affinity for BD2 over BD1. BET proteins interact with acetylated lysine-containing sequences to transcriptionally regulate several cellular processes. BRD4, a BET protein that mediates induction of Apolipoprotein A-I mRNA, can be disrupted at the binding site by RVX-208. This leads to altered transcription, resulting in increased ApoA-I production and high-density lipoprotein cholesterol levels, both of which are promising in the treatment of atherosclerosis and vascular inflammation. Recently, BRD4 inhibition has shown anti-viral activity and increased host resistance to several DNA and RNA viruses in vitro and in vivo, making BRD4 disruptors important compounds to study in relation to viral diseases.
References
[1] SARAH PICAUD. RVX-208, an inhibitor of BET transcriptional regulators with selectivity for the second bromodomain.[J]. Proceedings of the National Academy of Sciences of the United States of America, 2013, 110 49: 19754-19759. DOI:
10.1073/pnas.1310658110[2] KEVIN G MCLURE. RVX-208, an inducer of ApoA-I in humans, is a BET bromodomain antagonist.[J]. PLoS ONE, 2013: e83190. DOI:
10.1371/journal.pone.0083190[3] RVX-208: a small molecule that increases apolipoprotein A-I and high-density lipoprotein cholesterol in vitro and in vivo
[4] RAVI JAHAGIRDAR . A novel BET bromodomain inhibitor, RVX-208, shows reduction of atherosclerosis in hyperlipidemic ApoE deficient mice[J]. Atherosclerosis, 2014, 236 1: Pages 91-100. DOI:
10.1016/j.atherosclerosis.2014.06.008