Uses
Buprofezin is a contact and ingested insecticide, active against
Homoptera (whiteflies, leafhoppers, scale insects, etc.) in and on citrus,
cotton, cucumbers, tomatoes, sweet potatoes, rice, etc.
Uses
Buprofezin is a insecticide which works as a chitin synthesis inhibitor. Buprofezin is used to control stubborn rice pests and is effective in controlling green leaf hoppers and white back plant hoppe
rs for long durations.
Definition
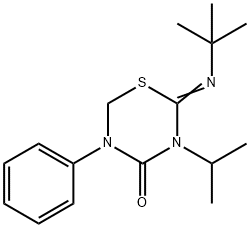
ChEBI: Buprofezin is a 2-(tert-butylimino)-5-phenyl-3-(propan-2-yl)-1,3,5-thiadiazinan-4-one in which the C=N double bond has Z configuration. It has a role as an insecticide and a member of homopteran inhibitor of chitin biosynthesis.
Hazard
Moderately toxic by ingestion. Low toxic-
ity by skin contact.
Agricultural Uses
Insecticide, Acaricide, Insect growth regulator: For insect control in food crops and greenhouse
ornamentals.
Trade name
APPLAUD®; NNI-750®
Pharmacology
The foregoing indicates that the modes of action of
buprofezin and benzoylureas could be similar or identical.
However, one differing biochemical effect of buprofezin is
inhibition of prostaglandin biosynthesis (33), a mechanism
that has been suggested as responsible for its ovicidal
activity. Subsequently, the in vitro and in vivo effects of
buprofezin were found to be strongly antagonized by 20-
hydroxyecdysone (34), which also affected prostaglandin
biosynthesis. Thus, inhibition of both prostaglandin and
chitin biosynthesis by buprofezin was prevented by 20-
hydroxyecdysone, so that both effects of the insecticide
are mediated via an effect on the hormone concentration
or its receptor. Consequently, buprofezin seems to inhibit
the drop in the 20-hydroxyecdysone titer that triggers
epidermal cell proliferation, old cuticle digestion, and new
cuticle deposition, but the detailed mechanism of this
action has yet to be established.
Metabolic pathway
Buprofezin gradually decomposes in soils under
flooded and upland conditions, with half-lives of
104 and 80 days, respectively. After 150 days, five
degradation products are identified as 2-tert-
butylimino-5-(4-hydroxyphenyl)-3-isopropylperhydro-
1,3,5-thiadiazin-4-one, 3-isopropyl-5-phenylperhydro-
1,3,5-thiadiazin-2,4-dione, 1-tert-butyl-3-ispropyl-5-
phenylbiuret, 1-isopropyl-3-phenylurea, and
phenylurea. As minor products, 2-tert-butylimino-5-
phenylperhydro-1,3,5-thiadiazin-4-one or buprofezin
sulfoxide are found in the flooded or in the upland
soils. Since neither formation of 14CO2 nor
hydroxylation is observed in the sterile soils,
buprofezin seems to have undergone complete
mineralization in soils under both conditions through
biological transformation by soil microorganisms.
Degradation
Buprofezin (1) was degraded under acidic conditions with half-lives
(DT
50) f 6-12 days (pH 4), 34 days (pH 6) and 65 days (pH 10) at 40 °C.
Opening of the thiadiazinanone ring appeared to be the primary hydrolytic
degradation pathway, to yield 1-tert-butyl-3-isopropyl-5-phenyl-2-
thiobiuret (2) and N-isopropyl-N-phenylurea (3) as major products.
Buprofezin is stable to aqueous photolysis. The estimated DT
50 of
buprofezin in distilled water when exposed to UV light was 39 days. A
more complex photodegradation pathway of buprofezin in methanol was
reported recently (Datta and Walia, 1997) with DT9 values of 4 hours and
15 days under UV and sunlight irradiation, respectively.