Description
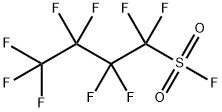
1,1,2,2,3,3,4,4,4-Nonafluorobutane-1-sulfonyl fluoride
(NfF) also called Nonafluorobutanesulfonyl fluoride is a versatile compound in organic synthesis. It can be
used as a fluoride source for the nucleophilic introduction
of fluorine, but it is also frequently applied as sulfonylation
reagent generating intermediates with strong electron withdrawing perfluorinated alkyl substituents.
Chemical Properties
Nonafluorobutanesulfonyl fluoride is a colorless, volatile liquid that is immiscible with water but soluble in common organic solvents. It is prepared by the electrochemical fluorination of sulfolane.
Uses
Nonafluorobutanesulfonyl fluoride can be used as a general-purpose diazotransfer reagent with high efficiency, wide substrate range, good shelf stability, relatively low cost, and easy purification. It is a favourable alternative to other commonly used diazotransfer reagents
[1]. In addition, Nonafluorobutanesulfonyl fluoride was used in the formation of a new dehydrated glycosylation scheme. In contrast to the main classical glycosylation reactions, this scheme glycosylation reaction is carried out under mildly alkaline conditions. In the absence of an external acceptor, the glycosyl hemiacetal undergoes self-condensation, providing the corresponding symmetric 1,1'- disaccharide in high yield
[2].
Uses
Benzynes were generated from o-(trimethylsilyl) phenols using nonafluorobutanesulfonyl fluoride (NfF) by a domino process.Nonafluorobutanesulfonyl fluoride (nonaflyl fluoride, C 4 F 9 SO 2 F, NfF) is the most widely used commercially available reagent for the synthesis of nonaflates.
Application
Perfluoro-1-butanesulfonyl fluoride (NfF) reacts with alcohols, including phenols, to yield nonafluorobutanesulfonate esters (nonaflates). Nonaflates can be used as electrophiles in several palladium-catalyzed cross coupling reactions and in Buchwald-Hartwig amination. NfF can also be used to prepare aryl nonaflates by reacting with corresponding aryloxysilanes in the presence of fluoride ion.
reaction suitability
reaction type: click chemistry
Synthesis
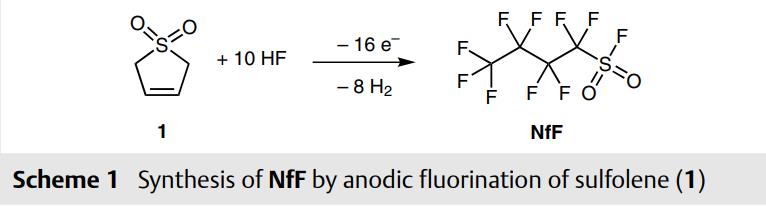
NfF(Nonafluorobutanesulfonyl fluoride) is produced on industrial scale by anodic fluorination of sulfolene (1, Scheme 1),3 therefore it is a fairly cheap reagent and commercially available from several suppliers. The compound is bench-stable and storable for years, nontoxic and easy to handle.
storage
Store in amber colored bottles protected from light; for prolonged storage, PVC bottles are recommended.
References
[1] JOSE LUIS CHIARA; José R S. Synthesis of α-Diazo Carbonyl Compounds with the Shelf-Stable Diazo Transfer Reagent Nonafluorobutanesulfonyl Azide[J]. Advanced Synthesis & Catalysis, 2011. DOI:10.1002/adsc.201000846.
[2] YU TANG; Biao Y; D Prabhakar Reddy. A dehydrative glycosylation protocol mediated by nonafluorobutanesulfonyl fluoride (NfF)[J]. Tetrahedron, 2021. DOI:10.1016/j.tet.2020.131800.