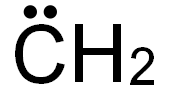
Carbene
Bezeichnung:Carbene
CAS-Nr47932-00-3
Englisch Name:Carbene
CBNumberCB91246530
Molgewicht0
MOL-Datei47932-00-3.mol
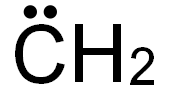
Bezeichnung:Carbene
CAS-Nr47932-00-3
Englisch Name:Carbene
CBNumberCB91246530
Molgewicht0
MOL-Datei47932-00-3.mol