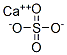
calcium sulfate
Bezeichnung:calcium sulfate
CAS-Nr99400-01-8
Englisch Name:calcium sulfate
CBNumberCB82130352
SummenformelCaO4S
Molgewicht136.14
MOL-Datei99400-01-8.mol
calcium sulfate physikalisch-chemischer Eigenschaften
| Schmelzpunkt | >300°C |
| storage temp. | Hygroscopic, Room Temperature, under inert atmosphere |
| Löslichkeit | Water (Slightly) |
| Aggregatzustand | Solid |
| Farbe | Off-White to Pale Grey |
| Wasserlöslichkeit | 2.4g/L |
| Solubility Product Constant (Ksp) | pKsp: 4.31 |
| Dielectric constant | 5.6(0.0℃) |
