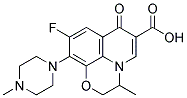
OFLOXACIN
Bezeichnung:OFLOXACIN
CAS-Nr83380-47-6
Englisch Name:OFLOXACIN
CBNumberCB2103600
SummenformelC18H20FN3O4
Molgewicht361.37
MOL-Datei83380-47-6.mol
Gefahreninformationscode (GHS)
-
Bildanzeige (GHS)


-
Alarmwort
Achtung
-
Gefahrenhinweise
H302:Gesundheitsschädlich bei Verschlucken.
H312:Gesundheitsschädlich bei Hautkontakt.
H317:Kann allergische Hautreaktionen verursachen.
H332:Gesundheitsschädlich bei Einatmen.
H334:Kann bei Einatmen Allergie, asthmaartige Symptome oder Atembeschwerden verursachen.
H341:Kann vermutlich genetische Defekte verursachen.
-
Sicherheit
P201:Vor Gebrauch besondere Anweisungen einholen.
P202:Vor Gebrauch alle Sicherheitshinweise lesen und verstehen.
P261:Einatmen von Staub vermeiden.
P264:Nach Gebrauch gründlich waschen.
P264:Nach Gebrauch gründlich waschen.
P270:Bei Gebrauch nicht essen, trinken oder rauchen.
P271:Nur im Freien oder in gut belüfteten Räumen verwenden.
P272:Kontaminierte Arbeitskleidung nicht außerhalb des Arbeitsplatzes tragen.
P280:Schutzhandschuhe/Schutzkleidung/Augenschutz tragen.
P281:Vorgeschriebene persönliche Schutzausrüstung verwenden.
P285:Bei unzureichender Belüftung Atemschutz tragen.
P301+P312:BEI VERSCHLUCKEN: Bei Unwohlsein GIFTINFORMATIONSZENTRUM/Arzt/... (geeignete Stelle für medizinische Notfallversorgung vom Hersteller/Lieferanten anzugeben) anrufen.
P302+P352:BEI BERÜHRUNG MIT DER HAUT: Mit viel Wasser/... (Hersteller kann, falls zweckmäßig, ein Reinigungsmittel angeben oder, wenn Wasser eindeutig ungeeignet ist, ein alternatives Mittel empfehlen) waschen.
P304+P340:BEI EINATMEN: Die Person an die frische Luft bringen und für ungehinderte Atmung sorgen.
P304+P341:BEI EINATMEN: Bei Atembeschwerden an die frische Luft bringen und in einer Position ruhigstellen, die das Atmen erleichtert
P308+P313:BEI Exposition oder falls betroffen: Ärztlichen Rat einholen/ärztliche Hilfe hinzuziehen.
P312:Bei Unwohlsein GIFTINFORMATIONSZENTRUM/Arzt/... (geeignete Stelle für medizinische Notfallversorgung vom Hersteller/Lieferanten anzugeben) anrufen.
P321:Besondere Behandlung
P322:Gezielte Maßnahmen
P330:Mund ausspülen.
P333+P313:Bei Hautreizung oder -ausschlag: Ärztlichen Rat einholen/ärztliche Hilfe hinzuziehen.
P342+P311:Bei Symptomen der Atemwege: GIFTINFORMATIONSZENTRUM/Arzt/... (geeignete Stelle für medizinische Notfallversorgung vom Hersteller/Lieferanten anzugeben) anrufen.
P363:Kontaminierte Kleidung vor erneutem Tragen waschen.
P405:Unter Verschluss aufbewahren.
P501:Inhalt/Behälter ... (Entsorgungsvorschriften vom Hersteller anzugeben) zuführen.
OFLOXACIN Chemische Eigenschaften,Einsatz,Produktion Methoden
-
S-Sätze Betriebsanweisung:
S22:Staub nicht einatmen.
S24/25:Berührung mit den Augen und der Haut vermeiden. -
Originator
Exocin,Allergan -
Definition
ChEBI: Ofloxacin is a racemate comprising equimolar amounts of levofloxacin and dextrofloxacin. It is a synthetic fluoroquinolone antibacterial agent which inhibits the supercoiling activity of bacterial DNA gyrase, halting DNA replication. It has a role as a DNA synthesis inhibitor, an antiinfective agent, an antibacterial drug, an EC 5.99.1.3 [DNA topoisomerase (ATP-hydrolysing)] inhibitor and a topoisomerase IV inhibitor. It is a quinolone antibiotic, a fluoroquinolone antibiotic and a racemate. It contains a levofloxacin and a dextrofloxacin. -
Manufacturing Process
20 g of 2,3,4-trifluoronitrobenzene was dissolved in 150 ml of dimethyl sulfoxide, and to this mixture a solution of 10% potassium hydroxide was added dropwise while keeping the temperature at 18° to 20°C. Then, the mixture was stirred for 2 hours at room temperature and one liter of water was added to this reaction mixture and the mixture was shaken with chloroform. The water layer was acidified with hydrochloric acid and was extracted with chloroform. The extract was washed with water and was dried, then chloroform layer was concentrated. The residue was purified by silica gel column chromatography to provide 5.8 g of 2,3-difluoro-6-nitrophenol as yellow oil.
7.9 g of the 2,3-difluoro-6-nitrophenol, 50.1 g of 1,2-dibromoethane and 18.7 g of potassium carbonate were added to 80 ml of dimethylformamide and the mixture was stirred for 2.5 hours at from about 80° to 100°C (bath temperature). The reaction mixture was concentrated to dryness in vacuo and the residue was distributed between ethyl acetate and water. The organic solvent layer was washed with water and was dried, then the solvent was evaporated. The residue was dissolved in benzene and was purified by silica gel column chromatography to provide 7.7 g of 2-(2-bromoethoxy)-3,4difluoronitrobenzene as light yellow oil.
1.74 g of this product was dissolved in 30 ml of methanol and a solution of 6.44 g of sodium dithionite dissolved in 15 ml of water was added thereto. The mixture was stirred for 1 hour at room temperature. Methanol was evaporated and the residue was extracted with chloroform. After the extract was washed with water and dried, the solvent was evaporated to provide 0.44 g of 2-(2-bromoethoxy)-3,4-difluoroaniline.
1.82 g of this product and 3.03 g of potassium carbonate were added to 10 ml of dimethylformamide and the mixture was stirred for 1 hour at from about 80° to 100°C (bath temperature). The reaction mixture was added to ice-cold water and was extracted with ethyl acetate. After the extract was washed with water and dried, the solvent was distilled off at room temperature to provide 1.21 g of 7,8-difluoro-2,3-dihydro-4H-[1,4]benzoxazine with m.p. 48°-54°C.
The mixture of 1.1 g of this product and 1.38 g of diethyl ethoxymethylenemalonate was stirred for 2 hours at from about 130° to 135°C (bath temperature). The ethanol produced was evaporated and 20 g of ethyl polyphosphate was added to the residue. Then the mixture was stirred for 1.5 hours at from about 140° to 145°C (bath temperature). The reaction mixture was added to ice-cold water and was extracted with chloroform. The extract was washed fully with water. After drying, the solvent was evaporated and the residue was recrystallized from ethyl acetate. 1.3 g of ethyl 9,10difluoro-7-oxo-2,3-dihydro-7H-pyrido[1,2,3-de][1,4]benzoxazine-6carboxylate was obtained as colorless needles with m.p. 265°-266°C.
1.15 g of this product was added to 12 ml of mixture of concentrated hydrochloric acid and acetic acid (1:4 by volume) and the mixture was stirred for 4 hours at 100° to 110°C (bath temperature). After cooling, the precipitated crystals were collected by filtration, washed with water, methanol and chloroform to give 0.78 g of 9,10-difluoro-7-oxo-2,3-dihydro-7Hpyrido[1,2,3-de][1,4]-benzoxazine-6-carboxylic acid as colorless needles with m.p. above 300°C.
1.0 g of 9,10-difluoro-3-methyl-7-oxo-2,3-dihydro-7H-pyrido[1,2,3de][1,4]benzoxazine-6-carboxylic acid and 2.85 g of N-methylpiperazine were added to 15 ml of dimethylsulfoxide. The mixture was stirred at a temperature of from about 100° to 110°C (bath temperature) for 12 hours and the reaction mixture was concentrated to dryness in vacuo and 40 ml of water was added to the residue. Then the product was extracted with chloroform. The extract was dried and concentrated to dryness in vacuo. The residue was recrystallized from ethanol to provide 550 mg of 9-fluoro-3-methyl-10-(4methyl-1-piperazinyl)-7-oxo-2,3-dihydro-7H-pyrido -
Therapeutic Function
Antibacterial
OFLOXACIN Anbieter Lieferant Produzent Hersteller Vertrieb Händler.
Global(51)Suppliers
-
Telefon +86-0371-86658258<br/>15093356674;
E-Mail laboratory@coreychem.com
-
Hefei TNJ Chemical Industry Co.,Ltd.
Telefon 0551-65418684<br/>+8618949823763
E-Mail sales@tnjchem.com
-
Hebei Duling International Trade Co. LTD
Telefon +8618032673083
E-Mail sales05@hbduling.cn
-
3B Pharmachem (Wuhan) International Co.,Ltd.
Telefon 821-50328103-801<br/>18930552037
E-Mail 3bsc@sina.com
-
XiaoGan ShenYuan ChemPharm co,ltd
Telefon 0712-0712-2580635<br/>15527768850
E-Mail 1791901229@qq.com
-
Beijing HuaMeiHuLiBiological Chemical
Telefon 010-56205725
E-Mail waley188@sohu.com
-
Hubei XinyuanShun Chemical Co., Ltd.
Telefon 13971561712, 13995564702, 027-50664929
E-Mail hbeixys2001@163.com
-
Telefon +86-21-5877 1921
E-Mail info@chemofchina.com
-
Telefon 020-81716320<br/>17788709170
E-Mail Sales@pipitech.com
-
PCL - Business, Laboratory Pharmaceutical
Telefon 19150123681
E-Mail Hu.xinyue@purechemland.com
1of2
