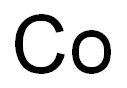
7440-48-4
| Name | Cobalt |
| CAS | 7440-48-4 |
| EINECS(EC#) | 231-158-0 |
| Molecular Formula | Co |
| MDL Number | MFCD00010935 |
| Molecular Weight | 58.93 |
| MOL File | 7440-48-4.mol |
Synonyms
Kobalt
COBALT
Aquacat
CO005085
CO004500
CO000070
CO000050
CO000295
CO004850
CO000292
CO004700
CO005120
CO000285
CO000290
CO000130
CO000100
CO000120
CO000281
CO000240
CO000210
CO005080
CO000291
CO000282
CO000270
CO000279
CO000160
CO000190
CO000201
CO005130
CO000280
CO004600
CO000110
CO000241
CO000080
CO005100
CO005090
CO005110
CO000140
CO000150
CO000180
CO000200
CO000170
CO000090
CO000060
Raney®
Cobalt-59
NCI-C60311
c.i. 77320
MATTCOBALT
Cobalt rod
CO STANDARD
supercobalt
cobalt fume
Cobalt wire
Cobalt foil
Cobalt slug
cobalt atom
Super cobalt
Cobalt powde
gcobalt atom
BRIGHTCOBALT
COBALT METAL
Cobalt, Lump
Cobalt Chunks
COBALT POWDER
Cobalt, pieces
COBALT STANDARD
ULTRAFINECOBALT
Cobaltwiremmdia
cobaltmetaldust
Cobalt Nanorods
Cobaltpowdermesh
COBALT,ELEMENTAL
Cobalt nanowires
Cobaltous powder
Cobalt solution
Cobaltfoil(99.9%)
Cobalt nanopowder
COBALT GRANULATED
Cobaltpowdermicron
cobalt,metalsponge
cobaltmetaldustand
Cobalt foil50x50mm
Cobalt powder 99+
COBALT 99.5%, SHOT
COBALT AA STANDARD
COBALTMETAL,PIECES
Cobalt(Metal)Powder
COBALT 99.9%, LUMPS
COBALT 99.5% POWDER
Cobaltpowder(99.8%)
CobaltpowderNmicron
COBALT ICP STANDARD
ELECTROLYTIC COBALT
raney(R) 2700 cobalt
Cobaltpowder(99.99%)
COBALT 99.5%, PIECES
Cobalt foil100x100mm
Cobalt(Metal)Cathode
COBALT FOIL, 3N :99%
Co Standard Solution
Cobalt Stock Solution
NANOPARTICULATECOBALT
COBALT, PIECES, 99.5%
Cobalt pieces (99.9+%
kobalt(german,polish)
cobaltmetaldustandfume
COBALT 99.999%, POWDER
Cobalt, Sponge, 99.99%
COBALT POWDER 99+ 100 G
COBALT ICP/DCP STANDARD
PLATING SOLUTION CO-801
PLATING SOLUTION CO-701
CloveOil(ForMicroscopy)
CobaltfoilNmmthickmmxmm
Cobalt, pellets, 1/4x1/2
Cobalt, pellets, 1/4x1/4
Cobalt, pellets, 1/8x1/4
Cobalt, pellets, 1/8x1/8
COBALT STANDARD SOLUTION
PLATING SOLUTION CO-100ES
COBALT POWDER: 99.8%, 2N8
COBALT POWDER: 99.99%, 4N
Cobalt, Quant Test Strips
LITHIUM 10,000PPM FOR ICP
COBALT, GRANULES, 99.995%
CobaltpiecesNrandompieces
Cobalt powder,sphere (Co)
COBALT CYCLOHEXYLBUTYRATE
Cobalt Rod/Φ8x100mm/99.99%
0.127MM THICK, 50MM X 50MM
Cobalt Rod/Φ2x100mm/99.95%
Cobalt Rod/Φ2x100mm/99.99%
Cobalt Rod/Φ6x100mm/99.99%
Cobalt,Powder,-180um,99.5%
Cobalt ISO 9001:2015 REACH
COBALT RANDOM PIECES: 99.9%
MOLYBDENUM 10,000PPM FOR ICP
cobaltmetaldustandfume(asco)
Cobalt, Powder, 99.5%, -45μm
Cobaltrod,6mmdia.x25mm,99,95%
COBALT, AAS STANDARD SOLUTION
Cobalt Metal, 400 Mesh, Powder
COBALT SINGLE ELEMENT STANDARD
cobalt coating quality balzers
Cobalt rod, 5mm (0.2 in.) dia.
COBALT METALLO-ORGANIC STANDARD
COBALT PLASMA EMISSION STANDARD
MAXIMUM RECOVERY DILUENT 20X9ML
COBALT STANDARD SOLUTION 500 ML
COBALT STANDARD SOLUTION 100 ML
Cobalt foil, 0.025mm (0.001 in.)
COBALT, PLASMA STANDARD SOLUTION
Cobalt Foil/0.10x50x50mm/99.99+%
Cobalt Foil/0.25x50x50mm/99.99+%
COBALT, ROD, 5.0MM DIAM., 99.95%
Cobalt Target/Φ25.4x3.2mm/99.99%
Cobalt rod, 12mm (0.47 in.) dia.
Cobalt Target//Φ25.4x6.3mm/99.99%
Cobalt Target//Φ50.8x3.2mm/99.99%
Cobalt Target//Φ50.8x6.3mm/99.99%
Cobalt foil (99.9%) (50mm x 50mm)
COBALT, FOIL, 0.5MM THICK, 99.95%
COBALT, FOIL, 0.1MM THICK, 99.95%
Cobalt Foil/ 0.05x50x50mm/99.99+%
COBALT, WIRE, 2.0MM DIAM., 99.9+%
COBALT, ROD, 5.0MM DIAM., 99.998%
COBALT, POWDER, < 2 MICRON, 99.8%
COBALT, WIRE, 0.5MM DIAM., 99.9+%
COBALT AA SINGLE ELEMENT STANDARD
COBALT ATOMIC ABSORPTION STANDARD
Cobalt Foil/ 0.50x50x50mm/99.99+%
Cobalt Foil/ 1.00x50x50mm/99.99+%
COBALT, WIRE, 1.0MM DIAM., 99.9+%
Cobaltgranules,99.5%(metalsbasis)
COBALT: 99.9%, POWDER, -150 MICRON
Cobalt, wire, 0.1mm dia., 99.9965%
COBALT ION CHROMATOGRAPHY STANDARD
COBALT, FOIL, 0.25MM THICK, 99.95%
COBALT, WIRE, 0.1MM DIAM., 99.995%
Cobalt wire, 1.5mm (0.06 in.) dia.
COBALT, FOIL, 1.0MM THICK, 99.99+%
COBALT, FOIL, 0.1MM THICK, 99.99+%
COBALT, WIRE, 0.5MM DIAM., 99.995%
Cobalt wire, 2.0mm (0.08 in.) dia.
Cobalt wire, 0.5mm (0.02 in.) dia.
Cobalt wire, 0.1mm (0.004 in.) dia.
Cobalt wire, 0.25mm (0.01 in.) dia.
Cobalt foil, 1.0mm (0.04 in.) thick
Cobalt foil, 1.5mm (0.06 in.) thick
Cobalt rod, 12.5mm (0.492 in.) dia.
Cobalt foil, 2.0mm (0.08 in.) thick
Cobalt foil, 0.5mm (0.02 in.) thick
COBALT, FOIL, 0.25MM THICK, 99.99+%
COBALT, WIRE, 0.25MM DIAM., 99.995%
COBALT ATOMIC SPECTROSCOPY STANDARD
COBALT, OIL BASED STANDARD SOLUTION
Cobalt ICP/DCP standard solution
Cobalt foil, 0.25mm (0.01 in.) thick
Cobalt foil, 0.1mm (0.004 in.) thick
Cobalt1,000μg/ginMineralOil,100Grams
Cobalt piece,8MM width ×2MM thickness
Cobalt foil, 0.05mm (0.002 in.) thick
Cobaltdisc;99.995%;2mmdiax0.5mmthick;
Cobalt Metal, 2 in. and Finer, Pieces
COBALT SINGLE ELEMENT PLASMA STANDARD
COBALT COATING QUALITY UMICORE TABLETS
Cobalt foil, thickness 0.1 mm, >=99.99%
Cobalt pieces, 99.5% trace metals basis
Cobalt pieces, 99.9% trace metals basis
COBALT ICP STANDARD TRACEABLE TO SRM FRO
COBALT AA/ICP CALIBRATION/CHECK STANDARD
Cobaltpowder,-150mesh,99.8%(metalsbasis)
COBALT POWDER: 99.8%, 2N8, 0.5-1.5 MICRON
Cobalt Tin (70:30wt%), powder, <150 micron
Cobalt, AAS standard solution, Co 1000μg/mL
Cobalt, plasma standard solution, Co 10μg/mL
Cobalt wire, 1.5mm (0.06 in.) dia., Annealed
COBALT PLASMA EMISSION SPECTROSCOPY STANDARD
Cobalt, 99.999%, (trace Metal basis), powder
Cobalt wire, 1.0mm (0.04 in.) dia., Annealed
Cobalt, foil, 1.0mm thick, 99.95% metals basis
Cobalt plasma standard solution, Co 10000μg/mL
Cobalt standard solution, 1 mg/ml Co in 2% HNO3
Cobalt, wire, 1.0mm diam., 99.995% metals basis
Cobalt (metal, dust, and fume as Co) and oxides
Cobalt, powder, <150 micron, 99.9+% metals basis
Cobalt rod, 5mm dia., 99.995% trace metals basis
Cobaltrod,12.5mm(0.492in)dia,99.95%(metalsbasis)
Cobalt, Reference Standard Solution, 1000ppm ±1%
COBALT, WIRE, DIAMETER 1 MM,PA CKAGE WIT H 1.4 G
COBALT ATOMIC ABSORPTION SINGLE ELEMENT STANDARD
Cobalt rod, 12mm dia., 99.995% trace metals basis
Cobalt AA Standard@1000 μg/mL in 2-5% Nitric Acid
Cobalt ICP Standard@1000 μg/mL in 2-5% Nitric Acid
Cobalt powder, -325 mesh, 99.5% trace metals basis
Cobalt rod, 5mm (0.2in) dia, 99.95% (metals basis)
Cobalt nanopowder, APS 5-15nm, 99.9% (metals basis)
Cobalt powder, -22 mesh, 99.995% trace metals basis
Cobalt powder, 1.6 micron, 99.5% trace metals basis
Cobalt rod, diam. 5.0 mm, 99.95% trace metals basis
Cobalt ICP Standard@10000 μg/mL in 2-5% Nitric Acid
Cobalt nanopowder, APS 25-30nM, 99.8% (Metals basis)
Cobalt foil, 0.127mm thick, 99.9% trace metals basis
Cobalt wire, diam. 1.0 mm, 99.995% trace metals basis
Cobalt, AAS standard solution, Specpure, Co 1000μg/ml
Cobalt ICP-MS Standard@100 μg/mL in 2-5% Nitric Acid
Cobalt ICP-MS Standard@1000 μg/mL in 2-5% Nitric Acid
Cobaltrod,5mm(0.2in)dia,Puratronic,99.995%(metalsbasis)
Cobalt, plasma standard solution, Specpure, Co 10g/ml
RANEY(R) 2700 COBALT, ACTIVE CATALYST, S LURRY IN WATER
Chemical Properties
| Appearance | Cobalt is a silver-gray to black, hard, brittle, magnetic metal. It is relatively rare; the important mineral sources are the arsenides, sulfides, and oxidized forms. It is generally obtained as a by-product of other metals, particularly copper. |
| Melting point | 1495°C |
| Boiling point | 2900 °C (lit.) |
| bulk density | 850kg/m3 |
| density | 1.03 g/mL at 25 °C |
| vapor pressure | 0Pa at 20℃ |
| storage temp. | no restrictions. |
| solubility | H2O: soluble |
| form | wire |
| color | Pink to red to violet |
| Specific Gravity | 8.9 |
| Flame Color | Silvery white |
| Stability: | Stable, but pyrophoric in air when finely divided. Incompatible with acetylene, hydrazinium nitrate, oxidizing agents, acids. |
| Resistivity | 6.24 μΩ-cm, 20°C |
| Electrical Conductivity | 1.7× 107S/m |
| Water Solubility | insoluble |
| Thermal Conductivity | 100 W/(m·K) |
| Merck | 13,2452 |
| Exposure limits | TLV-TWA 0.05 mg as Co/m3 (ACGIH) PEL-TWA: 0.05 mg as Co/m3 (NIOSH, OSHA) TLV-STEL 0.1 mg as Co/m3 (ACGIH) IDLH 20 mg as Co/m3 (NIOSH) . |
| InChI | 1S/Co |
| InChIKey | GUTLYIVDDKVIGB-UHFFFAOYSA-N |
| History | Cobalt occurs in the mineral cobaltite, smaltite, and erythrite, and is often associated with nickel, silver, lead, copper, and iron ores, from which it is most frequently obtained as a by-product. It is also present in meteorites. Important ore deposits are found in Congo-Kinshasa, Australia, Zambia, Russia, Canada, and elsewhere. The U.S. Geological Survey has announced that the bottom of the north central Pacific Ocean may have cobalt-rich deposits at relatively shallow depths in waters close to the Hawaiian Islands and other U.S. Pacific territories. Cobalt is a brittle, hard metal, closely resembling iron and nickel in appearance. It has a magnetic permeability of about two thirds that of iron. Cobalt tends to exist as a mixture of two allotropes over a wide temperature range; the β-form predominates below 400°C, and the α above that temperature. The transformation is sluggish and accounts in part for the wide variation in reported data on physical properties of cobalt. It is alloyed with iron, nickel and other metals to make Alnico, an alloy of unusual magnetic strength with many important uses. Stellite alloys, containing cobalt, chromium, and tungsten, are used for high-speed, heavy-duty, high-temperature cutting tools, and for dies. Cobalt is also used in other magnet steels and stainless steels, and in alloys used in jet turbines and gas turbine generators. The metal is used in electroplating because of its appearance, hardness, and resistance to oxidation. The salts have been used for centuries for the production of brilliant and permanent blue colors in porcelain, glass, pottery, tiles, and enamels. It is the principal ingredient in Sevre’s and Thenard’s blue. A solution of the chloride (CoCl2 · 6H2O) is used as sympathetic ink. The cobalt ammines are of interest; the oxide and the nitrate are important. Cobalt carefully used in the form of the chloride, sulfate, acetate, or nitrate has been found effective in correcting a certain mineral deficiency disease in animals. Soils should contain 0.13 to 0.30 ppm of cobalt for proper animal nutrition. Cobalt is found in Vitamin B-12, which is essential for human nutrition. Cobalt of 99.9+% purity is priced at about $250/kg. Cobalt-60, an artificial isotope, is an important gamma ray source, and is extensively used as a tracer and a radiotherapeutic agent. Single compact sources of Cobalt-60 vary from about $1 to $10/curie, depending on quantity and specific activity. Thirty isotopes and isomers of cobalt are known. |
| SMILES | [Co] |
| Hardness, Brinell | 125 |
| Hardness, Vickers | 253 |
| Modulus of Elasticity | 211 GPa |
| Poissons Ratio | 0.32 |
| Shear Modulus | 82.6 GPa |
| CAS DataBase Reference | 7440-48-4(CAS DataBase Reference) |
| IARC | 2B (Vol. 52) 1991, 2B (Vol. 86) 2006, 2A (Vol. 86) 2006 |
| NIST Chemistry Reference | Cobalt(7440-48-4) |
| EPA Substance Registry System | 7440-48-4(EPA Substance) |
Safety Data
| Symbol(GHS) |   GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302-H317-H319-H334-H341-H350-H360F-H412 |
| Precautionary statements | P273-P280-P301+P312-P302+P352-P305+P351+P338-P308+P313 |
| Hazard Codes | T,Xn,F |
| Risk Statements |
R45:May cause cancer.
R23/24/25:Toxic by inhalation, in contact with skin and if swallowed . R34:Causes burns. R53:May cause long-term adverse effects in the aquatic environment. R42/43:May cause sensitization by inhalation and skin contact . R40:Limited evidence of a carcinogenic effect. R36/37:Irritating to eyes and respiratory system . R36/38:Irritating to eyes and skin . R17:Spontaneously flammable in air. |
| Safety Statements |
S53:Avoid exposure-obtain special instruction before use .
S23:Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer) . S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice . S36/37/39:Wear suitable protective clothing, gloves and eye/face protection . S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible) . S61:Avoid release to the environment. Refer to special instructions safety data sheet . S37:Wear suitable gloves . S24:Avoid contact with skin . S22:Do not breathe dust . S36/37:Wear suitable protective clothing and gloves . S5:Keep contents under ... (appropriate liquid to be specified by the manufacturer) . |
| OEB | D |
| OEL | TWA: 0.05 mg/m3 |
| RIDADR | UN 3264 8/PG 3 |
| WGK Germany | 3 |
| RTECS | GF8750000 |
| TSCA | Yes |
| HS Code | 8105 20 00 |
| REACH Registrations | Active |
| HazardClass | 4.1 |
| PackingGroup | III |
| storage | Cobalt metal dust (powdered metal) should be stored in a cool, dry, well-ventilated area in tightly sealed containers that are labeled in accordance with OSHA standards. Containers of cobalt metal dust should be protected from physical damage and ignition sources, and should be stored separately from strong oxidizers. |
| Storage Class | 4.1B - Flammable solid hazardous materials |
| Hazard Classifications | Aquatic Chronic 4 Carc. 1B Eye Irrit. 2 Flam. Sol. 1 Muta. 2 Repr. 1B Resp. Sens. 1 Skin Sens. 1 |
| Safety Profile |
Confirmed carcinogen
with experimental neoplastigenic and
tumorigenic data. Poison by intravenous,
intratracheal, and intraperitoneal routes.
Moderately toxic by ingestion. Inhalation of
the dust may cause pulmonary damage. The
powder may cause dermatitis. Ingestion of
soluble salts produces nausea and vomiting
by local irritation. Powdered cobalt igmtes
spontaneously in air. Flammable when
exposed to heat or flame. Explosive reaction
with hydrazinium nitrate, ammonium nitrate
+ heat, and 1,3,4,7-tetramethylisoindole (at
39OOC). Ignites on contact with bromine
pentafluoride. Incandescent reaction with
acetylene or nitryl fluoride. See also
COBALT COMPOUNDS.
|
| Hazardous Substances Data | 7440-48-4(Hazardous Substances Data) |
| Toxicity |
LD50 orally in Rabbit: 6170 mg/kg
|
| IDLA | 20 mg Co/m3 |
Hazard Information
Chemical Properties
Cobalt is a silvery, bluish-white, odorless, and magnetic metal. The fume and dust of cobalt
metal is odorless and black. The appearance and odor of cobalt compounds and their dusts
and fumes vary with the compound. Cobalt metal in powdered form is incompatible with
fused ammonium nitrate, hydrozinium nitrate, and strong oxidizing agents and should be
avoided. It ignites on contact with bromide pentafl uoride. Powdered cobalt ignites spontaneously in air. Exposure to cobalt metal fume and dust can occur through inhalation,
ingestion, and eye or skin contact.
General Description
A black powder.
Reactivity Profile
Pyrophoric COBALT(7440-48-4) is a reducing agent. Decomposes acetylene in the cold as the metal becomes incandescent [Mellor 14:513(1946-1947]. Incompatible with oxidizing agents such as ammonium nitrate, bromine pentafluoride, and nitryl fluoride.
Air & Water Reactions
Burns brilliantly when exposed to air [Mellor 14:453(1946-1947)]. Insoluble in water.
Hazard
A possible carcinogen. Toxic by inhalation.
Dust is flammable. Asthma, myocardial effects, and
pulmonary function impairment.
Health Hazard
Acute exposure to cobalt metal, dust, and fume is characterized by irritation of the eyes
and, to a lesser extent, irritation of the skin. In sensitized individuals, exposure causes an
asthma-like attack, with wheezing, bronchospasm, and dyspnea. Ingestion of cobalt may
cause nausea, vomiting, diarrhea, and a sensation of heat.
Human exposures to cobalt and cobalt compounds cause cough, tight chest, pain in chest
on coughing, dyspnea, malaise, ache, chills, sweating, shivering, and aching pain in back
and limbs. After further days of exposures to high concentrations of cadmium, the worker
develops more severe pulmonary responses such as severe dyspnea, wheezing, chest pain
and precordial constriction, persistent cough, weakness and malaise, anorexia, nausea,
diarrhea, nocturia, abdominal pain, diffuse nodular fi brosis, respiratory hypersensitivity, asthma, abdominal pain, cardiomyopathy, lung damage, hemoptysis, prostration, and
death.
Cobalt metal, dust, and fume are pulmonary toxins and respiratory and skin sensitizers. Inhalation of cobalt metal fume and dust may cause interstitial fi brosis, interstitial
pneumonitis, myocardial and thyroid disorders, and sensitization of the respiratory tract
and skin. Chronic cobalt poisoning may also produce polycythemia and hyperplasia of the
bone marrow. Myocardial disorders have also been observed in cobalt production workers. Chronic exposure to cobalt metal, dust, or fume may cause respiratory or dermatologic
signs and symptoms. Following skin sensitization, contact with cobalt causes eruptions of
dermatitis in creases and on frictional surfaces of the arms, legs, and neck. Chronic cobalt
poisoning may cause polycythemia, hyperplasia of the bone marrow and thyroid gland,
pericardial effusion, and damage to the alpha cells of the pancreas.
Potential Exposure
Possible risk of forming tumors, Suspected reprotoxic hazard. Nickel-aluminumcobalt alloys are used for permanent magnets. Alloys with nickel, aluminum, copper, beryllium, chromium, and molybdenum are used in the electrical, automobile, and aircraft industries. Cobalt is added to tool steels to improve their cutting qualities and is used as a binder in the manufacture of tungsten carbide tools. Various cobalt compounds are used as pigments in enamels, glazes, and paints; as catalysts in afterburners; and in the glass, pottery, photographic, electroplating industries. Radioactive cobalt (60Co) is used in the treatment of cancer. Cobalt has been added to beer to promote formation of foam but cobalt acts with alcohol to produce severe cardiac effects at concentrations as low as 1.2-1.5 mg/L of beer. Cobalt is part of the vitamin B12 molecule and as such is an essential nutrient. The requirement of humans for cobalt in the form of vitamin B12 is about 0.13 μg/day.
Fire Hazard
Literature sources indicate that the dust of this chemical is flammable.
First aid
If this chemical gets into the eyes, remove any contact lenses at once and irrigate immediately for at least 15 minutes, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts the skin, remove contaminated clothing and wash immediately with soap and water. Seek medical attention immediately. If this chemical has been inhaled, remove from exposure, begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR if heart action has stopped. Transfer promptly to a medical facility. When this chemical has been swallowed, get medical attention. Give large quantities of water and induce vomiting. Do not make an unconscious person vomit. Medical observation is recommended for 24 to 48 hours after breathing overexposure, as pulmonary edema may be delayed. As first aid for pulmonary edema, a doctor or authorized paramedic may consider administering a drug or other inhalation therapy
Shipping
UN3189 Metal powder, self-heating, n.o.s., Hazard Class: 4.2; Labels: 4.2-Spontaneously combustible material
Description
Cobalt was discovered by George Brandt in 1737. Cobalt exists
in valence states from 0 to 5, with the most stable (+2 and +3)
being the most common. Although there is only one stable
isotope of cobalt, there are a number of unstable isotopes. Two
of these, cobalt-60 and cobalt-57, are in use commercially.
Cobalt-60 is used for cancer treatment and food irradiation.
Cobalt-57 has research applications.
Waste Disposal
Cobalt metal may be recovered from scrap and cobalt compounds from spent catalysts as alternatives to disposal.
Isotopes
There are 33 isotopes of cobalt, ranging from Co-48 to Co-75, with half-livesranging from a few nanoseconds to 5.272 years for cobalt-60. Cobalt-59 is the onlystable isotope that constitutes almost all (roughly 100%) of the element’s natural presence on Earth. All the other isotopes are radioactive and are created artificially in nuclearreactors or nuclear explosions.
Origin of Name
Cobalt was given the name kobolds (or kolalds, or kololos) by German
miners. It means “goblin” (see “History” for more on this story).
Occurrence
Cobalt is the 32nd most abundant element on Earth even though it makes up only 0.003%of the Earth’s crust. It is not found in the free metallic state, despite being widely distributedin igneous rocks as minerals. Its two most common mineral ores are cobaltite (CoAsS) anderythrite [Co3(AsO4)2]. These ores are placed in blast furnaces to produce cobalt arsenide(Co2As), which is then treated with sulfuric acid to remove the arsenic. Finally, the productcobalt tetraoxide (Co3O4) is reduced by heat with carbon (Co3O4 + C → 3Co + 2CO2), resulting in cobalt metal.Cobalt is also found in seawater, meteorites, and other ores such as linnaeite, chloanthite,and smaltite, and traces are found mixed with the ores of silver, copper, nickel, zinc, andmanganese. Cobalt ores are found in Canada and parts of Africa, but most of the cobalt usedin the United States is recovered as a by-product of the mining, smelting, and refining of theores of iron, nickel, lead, copper, and zinc.
Characteristics
Cobalt has the highest Curie point of any metal or alloy of cobalt. The Curie point is thetemperature at which an element will lose its magnetism before it reaches its melting point.Cobalt’s Curie point is 1,121°C, and its melting point is 1,495°C. About 25% of all cobaltmined in the world is used as an alloy with other metals. The most important is the alloyalnico, which consists of nickel, aluminum, and cobalt. Alnico is used to make powerful permanent magnets with many uses, such as CT, PET, and MRI medical instruments. It is alsoused for electroplating metals to give a fine surface that resists oxidation.
Definition
A lustrous silveryblue
hard ferromagnetic transition metal
occurring in association with nickel. It is
used in alloys for magnets, cutting tools,
and electrical heating elements and in catalysts
and some paints.
Definition
ChEBI: A cobalt group element atom that has atomic number 27.
Definition
cobalt: Symbol Co. A light-greytransition element; a.n. 27; r.a.m.58.933; r.d. 8.9; m.p. 1495°C; b.p.2870°C. Cobalt is ferromagneticbelow its Curie point of 1150°C.Small amounts of metallic cobalt arepresent in meteorites but it is usuallyextracted from ore deposits workedin Canada, Morocco, and Za?re. It ispresent in the minerals cobaltite,smaltite, and erythrite but also associatedwith copper and nickel as sulphidesand arsenides. Cobalt ores areusually roasted to the oxide and thenreduced with carbon or water gas.Cobalt is usually alloyed for use. Alnicois a well-known magnetic alloyand cobalt is also used to make stainlesssteels and in high-strength alloysthat are resistant to oxidation at hightemperatures (for turbine blades andcutting tools).
The metal is oxidized by hot airand also reacts with carbon, phosphorus,sulphur, and dilute mineralacids. Cobalt salts, usual oxidationstates II and III, are used to give abrilliant blue colour in glass, tiles,and pottery. Anhydrous cobalt(II)chloride paper is used as a qualitativetest for water and as a heat-sensitiveink. Small amounts of cobalt salts areessential in a balanced diet for mam-mals. Artificiallyproduced cobalt–60 is an importantradioactive tracer andcancer-treatment agent. The elementwas discovered by Georg Brandt(1694–1768) in 1737.
The metal is oxidized by hot airand also reacts with carbon, phosphorus,sulphur, and dilute mineralacids. Cobalt salts, usual oxidationstates II and III, are used to give abrilliant blue colour in glass, tiles,and pottery. Anhydrous cobalt(II)chloride paper is used as a qualitativetest for water and as a heat-sensitiveink. Small amounts of cobalt salts areessential in a balanced diet for mam-mals. Artificiallyproduced cobalt–60 is an importantradioactive tracer andcancer-treatment agent. The elementwas discovered by Georg Brandt(1694–1768) in 1737.
Reactions
Cobalt absorbs very little hydrogen even at high temperatures and nitrogen is practically
insoluble up to 1200°C. Finely divided cobalt is pyrophoric in air, but the massive metal
is scarcely attacked below 300°C. The oxide scale on cobalt heated in air or oxygen up to
900° consists of an outside layer of CO3O4 and a layer of CoO next to the metal ; above
900°, Co3O4 decomposes and the scale consists of CoO only. Cobalt reacts with many
non-metals when heated, e.g. the halogens, boron, sulphur, phosphorus, arsenic and
antimony, the reactions often proceeding with incandescence. Fluorine forms CoF3, while
the other halogens give the cobalt(II) halide.
Brand name
C.i. 77320;Cobalt-59;Impromin;Inter-con;Kometileneamin;Levacide-c;Orkomin;Panacur;Sofracaps;Tasvite;Trelenium.
World Health Organization (WHO)
WHO Comment(non-radioactive forms): The World Health Organization has no information further to the
above regarding preparations containing cobalt or to indicate that they are still
commercially manufactured.
Flammability and Explosibility
Notclassified
reaction suitability
reagent type: catalyst
core: cobalt
core: cobalt
Agricultural Uses
Cobalt (Co), a metallic element with an atomic weight of 58.94, is one of the transition elements belonging to the Group 9 (formerly Group VIII ) of the Periodic Table. However, in extremely low concentrations ranging from 0.1 to 10parts per billion (ppb), cobalt have been observed to improve growth, transpiration and photosynthesis of cotton, mustard and beans. Cobalt is required by symbiotic micro-organisms (e.g., rhizobia) for the fixation of elemental nitrogen through the formation of vitaminB12.
Cobalt forms a complex with nitrogen atoms of the porphyrin ring structure and provides a prosthetic group for association with nucleotides in vitamin B12 co-enzyme. This complex is called cobamide. Other cobalt functions include leghemoglobin metabolism and ribonucleotide reductase in Rhizobium, and activation of enolase, lecithinase and succinic kinase.
The concentration of cobalt in dry matter of plants ranges from 0.02 to 0.5 ppm. One ppb of cobalt in nutrient solution was found adequate for nitrogen fixation in alfalfa. The water content and catalase activity in leaves increased and the concentration of the cell sap decreased with cobalt application.
Cobalt content in soil is low and variable. In India, for instance, it ranges from 4 to 80ppm. The humus content of the soil influences the availability of cobalt in it. The nature of clay affects the absorption of cobalt from solutions, in the order muscovite > hematite > bentonite = kaolin. An increase in the pH of the soil decreases the availability of cobalt. Cobalt deficiency is more pronounced in coarse sandy soils and under high rainfall conditions. To overcome deficiency, cobalt fertilization with 100to 200g/ha as cobaltous sulphate (CoSO,) is recommended.
Cobalt forms a complex with nitrogen atoms of the porphyrin ring structure and provides a prosthetic group for association with nucleotides in vitamin B12 co-enzyme. This complex is called cobamide. Other cobalt functions include leghemoglobin metabolism and ribonucleotide reductase in Rhizobium, and activation of enolase, lecithinase and succinic kinase.
The concentration of cobalt in dry matter of plants ranges from 0.02 to 0.5 ppm. One ppb of cobalt in nutrient solution was found adequate for nitrogen fixation in alfalfa. The water content and catalase activity in leaves increased and the concentration of the cell sap decreased with cobalt application.
Cobalt content in soil is low and variable. In India, for instance, it ranges from 4 to 80ppm. The humus content of the soil influences the availability of cobalt in it. The nature of clay affects the absorption of cobalt from solutions, in the order muscovite > hematite > bentonite = kaolin. An increase in the pH of the soil decreases the availability of cobalt. Cobalt deficiency is more pronounced in coarse sandy soils and under high rainfall conditions. To overcome deficiency, cobalt fertilization with 100to 200g/ha as cobaltous sulphate (CoSO,) is recommended.
Industrial uses
Cobalt (symbol Co) is a lustrous, silvery-bluemetallic chemical element, resembling nickelbut with a bluish tinge instead of the yellow ofnickel. It is rarer and costlier than nickel andits price has varied widely in recent years.Although allied to nickel, it has distinctive differences.It is more active chemically thannickel. It is dissolved by dilute H2SO4, HNO3,or HCl acids, and is attacked slowly by alkalis.The oxidation rate of pure cobalt is 25 timesthat of nickel. Its power of whitening copperalloys is inferior to that of nickel, but smallamounts in Ni–Cu alloys will neutralize theyellowish tinge of the nickel and make themwhiter. The metal is diamagnetic like nickel, buthas three times the maximum permeability.Like tungsten, it imparts red-hardness to toolsteels. It also hardens alloys to a greater extentthan nickel, especially in the presence of carbon,and can form more chemical compoundsin alloys than nickel.
Its chemical properties resemble, in part,those of both nickel and iron. Cobalt is themetal with the highest Curie temperature(1121°C) and the lowest allotropic transformationtemperature (399°C). Below 421°C, cobaltis close-packed hexagonal; above, it is facecenteredcubic.
Its chemical properties resemble, in part,those of both nickel and iron. Cobalt is themetal with the highest Curie temperature(1121°C) and the lowest allotropic transformationtemperature (399°C). Below 421°C, cobaltis close-packed hexagonal; above, it is facecenteredcubic.
Biological Activity
Cobalt is a vital trace element in animal nutrition. Ruminants grazing upon cobaltdeficient pastures exhibit retarded growth, loss of appetite and anaemia ; rapid recovery from these symptoms occurs upon feeding the animals with a cobalt-supplemented diet. Cobalt salts are not therefore considered to be particularly toxic to animals, but to man they can in sufficiently large doses irritate the gastro-intestinal tract and cause nausea, vomiting and diarrhoea. Small amounts of cobalt, however, are invaluable in the treatment of pernicious anaemia. The discovery in 1926 of the antipernicious anaemia factor in liver led to the discovery in 1948 of vitamin B12, which was very soon after shown to contain cobalt.
Carcinogenicity
In mammalian cells in vitro cobalt compounds
have caused DNA strand breaks, sister
chromatid exchanges, and aneuploidy, but
not chromosomal aberrations.Cobalt salts
are generally nonmutagenic in prokaryotic
assays.
Environmental Fate
Cobalt most often depresses the activity of enzymes, including
catalase, amino levulinic acid synthetase, and P-450, enzymes
involved in cellular respiration. The Krebs citric acid cycle can
be blocked by cobalt resulting in the inhibition of cellular
energy production. Cobalt can replace zinc in a number of zincrequired
enzymes such as alcohol dehydrogenase. Cobalt can
also enhance the kinetics of some enzymes, such as heme
oxidase in the liver. Cobalt interferes with and depresses iodine
metabolism, resulting in reduced thyroid activity. Reduced
thyroid activity can lead to goiter.
Toxicity evaluation
The sources of cobalt in the environment are both natural and
synthetic (anthropogenic). Natural sources include soil,
seawater spray, volcanic eruptions, and forest fires. Anthropogenic
sources include combustion of fossil fuels, metal smelting,
sewage sludge, and processing of cobalt alloys. Cobalt is
found in the atmosphere in particulate form and returns to the
Earth’s surface through dry deposition and with rain or snow.
Once in surface water, cobalt generally moves into sediment.
Cobalt does not appear to biomagnify significantly in the
aquatic food chain. The cobalt that does accumulate in fish is
largely found in the nonedible parts of the fish. Seventy-nine
percent of the cobalt is transported by rivers globally and
precipitates in estuaries.
Under normal environmental conditions, cobalt is expected to bind strongly to soil; thus, migration through soil is very limited. Cobalt in soil can be taken up by plant roots and root vegetables, but does not translocate to the aboveground parts of plants.
Under normal environmental conditions, cobalt is expected to bind strongly to soil; thus, migration through soil is very limited. Cobalt in soil can be taken up by plant roots and root vegetables, but does not translocate to the aboveground parts of plants.
Incompatibilities
Cobalt metal dust/powder may spontaneously ignite on contact with air, when finely divided.Reacts with acids, strong oxidizers, ammonium nitrate,causing fire and explosion hazard. Can promote decomposition of various organic substances. Cobaltic oxide reactswith reducing agents; and violently with hydrogenperoxide
Toxics Screening Level
The initial threshold screening level (ITSL) for cobalt is 0.2 μg/m3 based on an 8 hour averaging time.
Questions And Answer
-
Chemical Properties
Cobalt is a silvery-gray brittle but hard metal, which is distributed widely in nature, including rocks, soil, plants, and animals. It is a metal with no odor and no vapor pressure at room temperature and is insoluble in water, except for ultrafine cobalt powder, which is water soluble. It is stable in air but chemically reactive with dilute acids and is a nonvolatile metal. Required in small amounts, cobalt is important for the sustenance of life. It is the only metal found in vitamins, most importantly as a part of vitamin B12 and is necessary in the formation of blood. Cobalt is a component of many naturally occurring minerals like sulfides, hydrates, and oxides. Cobalt compounds can occur in various oxidation states (0, +1, +2, +3, and +4) but the most common oxidation states are +2 and +3. Cobalt has one naturally occurring isotope, cobalt-59 and 26 radioactive isotopes.

Cobalt and its compounds are used as an important catalyst in various industrial processes. Cobalt catalysts are used in various processes such as a desulfurization catalyst for gas and oil and for polyesters, pottery, plastic, and detergent production. Cobalt is usually recovered as a byproduct of mining and refining of various minerals like copper and iron. It has the unique ability to be ductile as well as a malleable metal. This property allows it to be transformed into both thin wires and thin sheets. ; -
Physical properties
Cobalt is a hard magnetic metal, like iron and nickel. It has the properties of both ductility as well as malleability. Due to this property, it can be molded into wires and compressed into sheets. Cobalt occurs in two allotropic forms, hexagonal and cubic. The hexagonal form is more stable than the cubic form at room temperature. The boiling point of cobalt is about 2927 °C (5301°F) and the melting point is about 1490 °C (2723°F). Cobalt as a metal is mildly reactive with oxygen and will burn only in powder form. It is soluble in dilute acids but is water soluble only in ultrafine powder form (Kyono et al., 1992).; -
Discovery
Cobalt(Co) is a metallic chemical element, specifically number 27 on the Periodic Table of the Elements. Cobalt was first discovered by George Brandt in 1739, a Swedish chemist, but it was not until 1780 that cobalt was confirmed as an element by Torbern Bergman. Cobalt has a long history of being mined, from which its name is a relic. Cobalt is derived from the German word kobold, which translates into“goblin”or “evil spirit,”as it was obtained as a byproduct of silver mining in Germany. When the cobalt-containing ore was smelted to isolate the silver, toxic arsenic-trioxide (AsO3) gas was released, which the miners attributed to the cobalt rather than the arsenic contained in the ore. Despite its inauspicious beginnings, cobalt has become an integral part of our technologically advanced world. ; -
History
The metal was isolated by Brandt in 1735 and confirmed as an element by Bergman in 1780. Cobalt is widely distributed in nature, but in small concentrations. Its concentration in the earth’s crust is estimated to be about 0.0025% and in the sea water is about 0.02 µg/L. Most cobalt found on earth is diffused into the rocks. It also is found in coal and soils, and at trace concentations in animals and plants. It is an essential element for plants and animals (as vitamin B12). Its absence in animals can cause retarded growth, anemia and loss of apetite. The element has been detected in meteorites and in the atmospheres of the sun and other stars. ; -
Uses
Cobalt has been in use as a coloring agent for glass since ancient times. The most imporant use of cobalt is in the manufacture of various wearresistant and superalloys. Its alloys have shown high resistance to corrosion and oxidation at high temperatures. They are used in machine components. Also, certain alloys are used in desulfurization and liquefaction of coal and hydrocracking of crude oil shale. Cobalt catalysts are used in many industrial processes. Several cobalt salts have wide commercial applications Cobalt oxide is used in glass to impart pink or blue color. Radioactive cobalt–60 is used in radiography and sterilization of food. ; -
Production Methods
Cobalt is obtained from its ores, which are mostly sulfide, arsenic sulfide or oxide in nature. The finely ground ore is subjected to multistep processing, depending on the chemical nature of the ore.
When the sulfide ore carrollite, CuS•Co2S3, is the starting material, first sulfides are separated by flotation with frothers. Various flotation processes are applied. The products are then treated with dilute sulfuric acid producing a solution known as copper-cobalt concentrate. This solution is then electrolyzed to remove copper. After the removal of copper, the solution is treated with calcium hydroxide to precipitate cobalt as hydroxide. Cobalt hydroxide is filtered out and separated from other impurities. Pure cobalt hydroxide then is dissolved in sulfuric acid and the solution is again electrolyzed. Electrolysis deposits metallic cobalt on the cathode.
Production of cobalt in general is based on various physical and chemical processes that include magnetic separation (for arsenic sufide ores), sulfatizing roasting (for sulfide ores), ammoniacal leaching, catalytic reduction, and electrolysis.
Finely divided cobalt particles can be prepared by reduction of cobalt(II) chloride by lithium naphthalenide in glyme. ;
Supplier
Shanghai Xianxin New Materials Technology Co. Ltd.
Telephone021-34790236 13056781586
Websitehttp://www.xxnano.com
Shanghai Yaotian New Material Technology Co., LTD
Telephone021-64110338 18939791287
Websitehttps://www.chemicalbook.com/ShowSupplierProductsList1440513/0_EN.htm
ZHEJIANG YAMEI NANO TECHNOLOGY CO., LTD.
Telephone400-8200386 13456373028
Websitehttps://www.ameinano.com
J & K SCIENTIFIC LTD.
Telephone18210857532; 18210857532
Websitehttps://www.jkchemical.com
Alfa Aesar
Telephone400-6106006
Websitehttp://chemicals.thermofisher.cn
Beijing HwrkChemical Technology Co., Ltd
Telephone89508211 18501085097
Websitehttp://www.hwrkchemical.com
Energy Chemical
Telephone021-021-58432009 400-005-6266
Websitehttp://www.energy-chemical.com
JinYan Chemicals(ShangHai) Co.,Ltd.
Telephone13817811078
Websitehttp://www.jingyan-chemical.com/
Shanghai Hanhong Scientific Co.,Ltd.
Telephone021-54306202 13764082696
Websitehttps://www.hanhongsci.com
Shandong Xiya Chemical Co., Ltd
Telephone13355009207 13355009207
Websitehttp://www.xiyashiji.com
Sichuan Kulinan Technology Co., Ltd
Telephone400-1166-196 18981987031
Websitehttp://www.hx-r.com/
Tianjin heowns Biochemical Technology Co., Ltd.
Telephone400 638 7771
Websitehttp://www.heowns.com
Sinopharm Chemical Reagent Co,Ltd.
Telephone86-21-63210123
Websitehttp://www.reagent.com.cn
Beijing Yan Nuo XinCheng Chemical Co., Ltd.
Telephone86-10-82830276
Websitehttps://www.chemicalbook.com/ShowSupplierProductsList15334/0_EN.htm
Maya High Purity Chemicals
Telephone+86 (573) 82222445 (0)18006601000 452520369
Websitehttps://www.chemicalbook.com/ShowSupplierProductsList15221/0_EN.htm
Spectrum Chemical Manufacturing Corp.
Telephone021-021-021-67601398-809-809-809 15221380277
Websitehttps://www.spectrumchemical.com/OA_HTML/index.jsp?minisite=10020&respid=22372&language=US
Chengdu Ai Keda Chemical Technology Co., Ltd.
Telephone4008-755-333 18080918076
Websitehttp://www.aikeshiji.com
Thermo Fisher Scientific
Telephone800-810-5118
Websitehttp://www.thermo.com.cn
1of4
PROMPT×
PROMPT
The What'sApp is temporarily not supported in mainland China
The What'sApp is temporarily not supported in mainland China
Cancel
Determine