Description
Repaglinide is a metaglitinide antidiabetic agent that blocks ATP-dependent potassium (K
ir6) channels in pancreatic β-cells (K
d = 0.42 nM for the sulphonylurea receptor SUR1 when co-expressed with K
ir6.2).
1 In vivo, repaglinide lowers blood glucose in fasted rats and dogs (ED
50s = 10 and 28.3 μg/kg, respectively).
2 Formulations containing repaglinide have been used to control blood sugar levels in patients with type 2 diabetes.
Description
Repaglinide was marketed in the US as an orally active hypoglycemic
agent in patients with a type Ⅱ diabetes mellitus, to lower blood glucose in
synergistic combination with metformin, when hyperglycemia cannot be
controlled by diet, exercise or metformin alone. Chiral (S)-repaglinide can be
synthesized in several ways, each involving a stereoselective reduction of an
imine or enamine group. Repaglinide is a nonsulfonylurea belonging to the
meglitinide family, with an insulin-releasing effect mediated by pancreatic betacells,
by closing the ATP-sensitive K+ channels that, in turn, increases the
cytosolic concentration of Ca++. In several animal models, repaglinide was more
efficient than glibenclamide as a dose-dependant promoter of insulin release,
but its mechanism of action is probably slightly different. Repaglinide has a rapid
onset of action, a short duration of action and a reduced risk of hypoglycemia
compared to glyburide.
Originator
Boehringer Ingelheim (Germany)
Definition
ChEBI: Repaglinide is a member of piperidines.
Brand name
Prandin (Novo Nordisk).
General Description
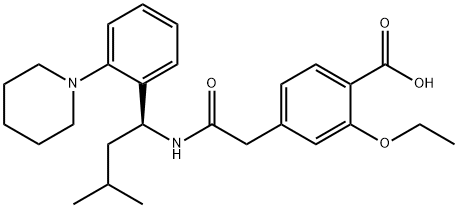
Repaglinide is 2-ethoxy-4-[2-[[(1S)-3-methyl-1-[2-(1-piperidinyl)phenyl]butyl]amino]-2-oxoethyl]benzoic acid(Prandin); approvals for generics are pending. Combinationsare available with metformin in the United States(Prandimet), and the drug may also be coprescribed with oneof the thiazolidinediones (typically pioglitazone or rosiglitazone;see previous discussion). To establish the most clinicallyvaluable dose, the patient is titrated while monitoringblood glucose levels and hemoglobin glycosylation (HbA1c)as an index of longer-term overall control.
General Description
Repaglinide, (+)-2-ethoxy-4-[N-[3-methyl-1(S)-[2-(1-piperidinyl) phenyl]butyl]carbamoyl-methyl]benzoicacid (Prandin), represents a new class of nonsulfonylureaoral hypoglycemic agents. With a fast onset and ashort duration of action, the medication should be taken withmeals. It is oxidized by CYP 3A4, and the carboxylic acidmay be conjugated to inactive compounds. Less than 0.2%is excreted unchanged by the kidney, which may be an advantagefor elderly patients who are renally impaired. Themost common side effect involves hypoglycemia, resultingin shakiness, headache, cold sweats, anxiety, and changes inmental state.
Biological Activity
K ATP channel blocker that binds with high affinity for SUR1 when co-expressed with Kir6.2 (K d = 0.42 nM). Antidiabetic glucose regulator with hypoglycaemic effect in vivo .
Biochem/physiol Actions
Repaglinide is a potent short-acting insulin secretagogue that acts by closing ATP-sensitive potassium (KATP) channels in the plasma membrane of the pancreatic beta cell. It represents a new class of insulin secretagogues, structurally unrelated to sulphonylureas, which were developed for the treatment of type 2 diabetes.
Clinical Use
Repaglinide is a nonsulfonylurea insulin secretagogue that was introduced in the United States in 1998 for type 2 diabetes.
Drug interactions
Potentially hazardous interactions with other drugs
Antibacterials: effects enhanced by clarithromycin
and possibly trimethoprim - avoid with
trimethoprim; hypoglycaemic effect antagonised by
rifampicin.
Antifungals: effect possibly enhanced by itraconazole.
Ciclosporin: may increase repaglinide concentration,
possibly enhanced hypoglycaemic effect.
Clopidogrel: avoid concomitant use if possible due to
increased repaglinide exposure.
Cytotoxics: avoid with lapatinib.
Lipid-lowering agents: increased risk of severe
hypoglycaemia with gemfibrozil - avoid.
Metabolism
Repaglinide appears to be a substrate for active hepatic
uptake by the organic anion transporting protein
OATP1B1, and undergoes almost complete hepatic
metabolism involving the cytochrome P450 isoenzymes
CYP2C8 and CYP3A4. The glucuronidation of
repaglinide is thought to involve uridine diphosphate
glucuronosyltransferase (UGT) enzymes, particularly
UGT1A1.
The metabolites, which are inactive, are excreted in the
bile.
References
[1] ANN MARIA K HANSEN. Kir6.2-dependent high-affinity repaglinide binding to β-cell KATP channels[J]. British Journal of Pharmacology, 2009, 144 4: 551-557. DOI:
10.1038/sj.bjp.0706082[2] MICHAEL MARK Wolfgang G. Hypoglycaemic effects of the novel antidiabetic agent repaglinide in rats and dogs[J]. British Journal of Pharmacology, 2009, 121 8: 1597-1604. DOI:
10.1038/sj.bjp.0701307