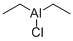-
外観
無色~ほとんど無色透明液体
-
溶解性
水, アルコールにより分解。炭化水素, エーテルに易溶。ヘキサン及びベンゼンと任意の割合で混和するが、水及びエタノールには分解する。
-
解説
(C2H5)2AlCl(120.56).有機アルミニウム化合物の一つ.AlとMgの合金または粉末混合物と塩化エチルとの反応で生成する.そのほかに(C2H5)5Al2Cl3のナトリウムアマルガムによる分解でも得ることができる.無色の液体.融点-74 ℃,沸点214 ℃.d2540.958.有機溶媒に可溶.液体では二量体として存在している.空気や水によりはげしく発火して分解する.酸素と反応してクロロジエトキシアルミニウム(C2H5O)2AlClを,二酸化炭素と反応してプロピオン酸を生じる.チーグラー-ナッタ触媒の一成分であり,またエチル化剤としても重要である.[CAS 96-10-6]
森北出版「化学辞典(第2版)
-
用途
有機合成用の金属触媒。オレフィン類の重合触媒。
-
使用上の注意
本品を採取する場合は、乾燥して窒素を充填した注射器等を用いること。不活性ガス封入
-
化学的特性
colorless solution
-
調製方法
Ethylaluminum sesquichloride (26.5 kg) added to a nitrogen-purged reactor, was heated to 175 ℃. Then, while the mixture was stirred vigorously, 1.1 kg sodium was added over a 30 min period; the mixture was further heated at 155 – 190 ℃ for 60 min. The diethylaluminum chloride product distilled from the reactor at 100 – 161 ℃ (1.3 – 6.1 kPa). In this example, an excess of ethylaluminum sesquichloride was employed to facilitate draining the voluminous byproduct salt and aluminum solids from the reactor. In an alternate approach, a heavy hydrocarbon oil, added prior to reaction, may be employed to remove the solids in slurry form, permitting the use of a stoichiometric ratio of ethylaluminum sesquichloride and sodium reactants.
-
一般的な説明
Colorless liquid. Dangerous fire and explosion hazard. Used as an intermediate in production of organometallics.
-
空気と水の反応
Pyrophoric in air [Hawley]. Reacts violently with water, Rose(1961).
-
反応プロフィール
Organometallics, such as DIETHYLALUMINUM CHLORIDE, are reactive with many other groups. Incompatible with acids and bases. Organometallics are good reducing agents and therefore incompatible with oxidizing agents. Often reactive with water to generate toxic or flammable gases. Organometallics containing halogens (fluorine, chlorine, bromine, iodine) bonded to the metal typically with generate gaseous hydrohalic acids (HF, HCl, HBr, HI) with water.
-
職業ばく露
These materials are used as components of olefin polymerization catalysts. The reader is referred to the entry on “Aluminum alkyls” for additional information on this entry. The aluminum alkyl halides parallel very closely the aluminum alkyls
-
輸送方法
UN3052 Spontaneously combustible. Water reactive releasing large quantities of toxic and deadly hydrogen gas. (Note: this number does not appear in the 49/CFR HazMat tables)
-
純化方法
Distil it from excess dry NaCl (to remove ethyl aluminium dichloride) in a 50-cm column containing a heated nichrome spiral. [Beilstein 4 IV 4403.]
-
不和合性
The aluminum alkyl halides are strong reducing agents; they react—possibly violently—with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. These chemicals react violently with nitromethaneEthylaluminum sesquichloride reacts explosively with carbon tetrachloride at room temperature. This chemical reacts violently with water, forming corrosive hydrogen chloride and flammable ethane gas. Diethylaluminum chloride may form an explosive product with chlorine azide.