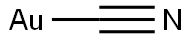-
外観
黄色の粉末
-
溶解性
水に不溶。水、エタノール、エーテルに不溶。アンモニア、水酸化アルカリ、シアン化アルカリ、チオ硫酸ナトリウム又は硫化アンモニウム溶液に可溶。
-
解説
シアン化金(Ⅰ)(gold(Ⅰ) cyanide):AuCN(222.98).ジシアノ金(Ⅰ)酸カリウムK[Au(CN)2]とHClとの水溶液を50 ℃ に加熱すると得られる.淡黄色の結晶.鎖状構造(…Au-CN-Au…).密度7.14 g cm-3.水に不溶.乾燥空気中では安定だが,湿った空気中,光照射,加熱などで分解して,Auとジシアン (CN)2 になる.NH3,KOH,KCNなどの水溶液に可溶.
-
用途
メッキ材料。
-
使用上の注意
空気中で安定であるが、湿った状態で光にさらすと分解する。
-
化学的特性
yellow powder(s); hexagonal; odorless; iridescent in sunlight; slowly decomposes in presence of moisture [MER06]
-
物理的性質
Yellow hexagonal crystals; odorless; density 7.14 g/cm3; decomposes slowly in the presence of moisture or decomposes on heating; insoluble in water, alcohol and ether; also insoluble in dilute acids; soluble in aqueous solutions of potassium-, or sodium cyanide, ammonia, and aqua regia.
-
使用
Electrolyte in the electroplating industry
-
製造方法
Gold(I) cyanide may be prepared by boiling sodium aurus cyanide, Na[Au(CN)2] with hydrochloric acid:Na[Au(CN)2] + HCl AuCN + HCN + NaCl
The complex cyanide, Na[Au(CN)2] is made by dissolving gold in a dilute solution of sodium cyanide in the presence of air; or by dissolution of a gold anode in a solution of sodium cyanide during electrolysis. The solution is evaporated to separate the complex, Na[Au(CN)2], which is purified by recrystallization from water. Potassium cyanide may be used instead of sodium cyanide to prepare gold(I) cyanide.
-
危険性
Toxic material.
-
純化方法
The lemon yellow powder is sparingly soluble in H2O and EtOH but soluble in aqueous NH3. It is obtained by heating H[Au(CN)2] at 110o. Wash it well with H2O and EtOH and dry it at 110o. It has an IR band at max 2239 cm -1 typical for C� stretching vibration. [Glemser & Sauer Handbook of Preparative Inorg anic Chemistry (Ed. Brauer) Academic Press Vol II p 1064 1965.] CARE: may evolve HCN.