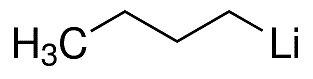
-
外観
うすい黄赤色~黄色~緑色透明液体~濁った液体
-
溶解性
水, アルコールにより分解。炭化水素, エーテルに易溶。炭化水素及びエーテル類に極めて溶けやすく、水によって急速に分解し、エタノール又は酸によって激しく分解する。
-
用途
有機合成試薬。
-
用途
共役ジエンのアニオン重合開始剤、飽和および不飽和カルボニル化合物のアルキル化反応などに用いられる。
-
使用上の注意
不活性ガス封入
-
化学的特性
n-Butyllithium is a colorless crystalline solid or clear yellow solution. Due to its high reactivity and extreme solubility, even in its impurities, it is commonly thought to be a viscous liquid.
-
使用
n-Butyllithium is used as a polymerization initiator in the production of elastomers like polybutadiene and styrene- butadiene- styrene. As a strong base, it is utilized in the synthesis of organic compounds. It undergoes thermal decomposition to prepare 1-butene.
-
製造方法
Industrially, n-butyllithium is produced by the
reaction of n-butyl chloride with lithium metal dispersion in various hydrocarbon solvents.
Hexane is the most commonly used solvent. Up to one-half of the lithium is replaced with
sodium in order to lower the cost of the butyllithium and increase the reactivity of the dispersion.
The reaction is carried out below the boiling point of the solvent.
The laboratory procedure is essentially the same except that pentane and diethyl ether
are two of the more popular solvents. The preparation runs smoothly in ether at lower
temperatures but the product must either be used immediately or kept refrigerated due to
the rapid cleavage of ether by n-butyllithium.
-
燃焼性と爆発性
The risk of fire or explosion on exposure of butyllithium solutions to the atmosphere
depends on the identity of the organolithium compound, the nature of the solvent,
the concentration of the solution, and the humidity. t-Butyllithium solutions are the
most pyrophoric and may ignite spontaneously on exposure to air. Dilute solutions
(1.6 M, 15% or less) of n-butyllithium in hydrocarbon solvents, although highly
flammable, have a low degree of pyrophoricity and do not spontaneously ignite.
Under normal laboratory conditions (25 °C, relative humidity of 70% or less),
solutions of -20% concentration will usually not ignite spontaneously on exposure to
air. More concentrated solutions of n-butyllithium (50 to 80%) are most dangerous
and will immediately ignite on exposure to air. Contact with water or moist materials
can lead to fires and explosions, and the butyllithiums also react violently with
oxygen.
-
貯蔵
In
particular, butyllithium should be stored and handled in areas free of ignition
sources, and containers of butyllithium should be stored under an inert atmosphere.
Work with butyllithium should be conducted in a fume hood under an inert gas such
as nitrogen or argon. Safety glasses, impermeable gloves, and a fire-retardant
laboratory coat are required.
-
不和合性
The butyllithiums are extremely reactive organometallic compounds. Violent
explosions occur on contact with water with ignition of the solvent and of the butane
produced. t-Butyllithium will ignite spontaneously in air. The butyllithiums ignite on
contact with water, carbon dioxide, and halogenated hydrocarbons. The
butyllithiums are incompatible with acids, halogenated hydrocarbons, alcohols, and
many other classes of organic compounds.
-
廃棄物の処理
Excess butyllithium solution can be destroyed by dilution with hydrocarbon solvent to a concentration of
approximately 5 wt %, followed by gradual addition to water with vigorous stirring under an inert
atmosphere. Alternatively, the butyllithium solution can be slowly poured (transfer by cannula for s- or tbutyllithium)
into a plastic tub or other container of powdered dry ice.
The residues from the above procedures and excess butyllithium should be placed in an appropriate
container, clearly labeled, and handled according to your institution's waste disposal guidelines.