-
外観
うすい橙色~赤褐色又は黒褐色、結晶~粉末
-
解説
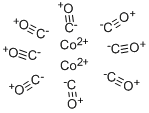
ジコバルトオクタカルボニルCo2(CO)8の組成をもつ二核錯体.テトラカルボニルコバルトヒドリドを常温で熱分解すると得られる.暗褐色の結晶.昇華精製により橙色の結晶が得られる.蒸気圧は15 ℃ で9.33 Pa.融点51 ℃.これより上の温度では分解して [Co(CO)3]4 になる.この錯体はヒドロホルミル化反応(オキソ合成)の触媒となる.
-
使用上の注意
アルゴン封入
-
化学的特性
Cobalt carbonyl is a pyrophoric (spontaneously flammable in air), red-orange (when pure) to dark-brown crystalline solid.
-
物理的性質
Orange crystals; density 1.78 g/cm3; melts at 5l°C; decomposes above this temperature; insoluble in water; soluble in most organic solvents including alcohol, ether, carbon disulfide.
-
使用
- Cobalt carbonyl [Co2(CO)8] is commonly used as a catalyst in the hydroformylation (oxo reaction) of alkenes.
- Along with pyridine, it can be used as a catalyst in the carboxylation of alkenes into corresponding acids and esters.
- It is employed as a key precursor in the preparation of cobalt platinum (CoPt3), cobalt sulfide (Co3S4) and cobalt selenide (CoSe2) nanocrystals.
- It is also used as a reagent in Pauson-Khand cyclizations and Nicholas reaction.
-
製造方法
Cobalt octacarbonyl is prepared by the reaction of finely divided cobalt with carbon monoxide under pressure:
2Co + 8CO → Co2(CO)8
The compound may be prepared in a similar way from cobalt(II) iodide. Also, it may be prepared by thermal decomposition of cobalt carbonyl hydride:
2HCo(CO)4 → Co2(CO)8 + H2
-
危険性
Toxic by ingestion and inhalation.
-
健康ハザード
Dicobalt octacarbonyl exhibits moderate toxicityby inhalation route and somewhatlower toxicity by intraperitoneal and oralroutes. However, it is much less toxicthan nickel tetracarbonyl or iron pentacarbonyl.A 2-hour LC50 value in mice isreported as 27 mg/m3 (Lewis 1996). Anoral LD50 value in rats is within the rangeof 750–800 mg/kg. It decomposes, evolvingtoxic carbon monoxide.
-
安全性プロファイル
Poison by inhalation
and intraperitoneal routes. Questionable
carcinogen. Decomposes in air to form a
product that ignites spontaneously in air.
"hen heated to decomposition it emits
acrid smoke and fumes. See also
CARBONYLS and COBALT
COMPOUNDS.
-
職業ばく露
This material is used as a catalyst for a number of reactions. It is also used in antiknock gasoline and for high-purity cobalt salts.
-
輸送方法
UN3124 Toxic solids, self-heating, n.o.s., Hazard Class: 6.1; 6.1-Poisonous materials, 4.2-Spontaneously combustible material. Technical Name Required. UN3190 Self-heating solid, inorganic, Hazard Class: 4.2; Labels: 4.2-Spontaneously combustible material, Technical Name Required. UN1325 Flammable solid, organic, n.o.s. Hazard Class: 4.1; Labels: 4.1-Flammable solid
-
純化方法
It forms orange-brown crystals on recrystallisation from n-hexane under a carbon monoxide atmosphere [Ojima et al. J Am Chem Soc 109 7714 1987; see also Hileman in Preparative Inorganic Reactions, Ed. Jolly, Vol 1 p 101 1987].
-
不和合性
Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. Decomposes on exposure to air or heat (@ ~52°C) producing toxic fumes of cobalt and oxides of carbon