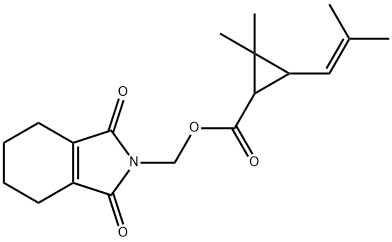
-
外観
白色〜わずかにうすい黄色, 結晶性粉末〜粉末
-
用途
医薬部外品原料(防疫用殺虫剤),殺菌剤(失効農薬)
-
効能
殺虫薬
-
説明
Form: Colorless crystals with slight pyrethrum-like odor
-
化学的特性
white crystals or powder
-
使用
Tetramethrin is a synthetic pyrethroid pesticide used in large-scale commercial agricultural applications as well as in consumer products for domestic purposes.
-
一般的な説明
Colorless crystals with slight odor. Non corrosive. Used as an insecticide.
-
空気と水の反応
Hydrolysis occurs with strong acid or base.
-
反応プロフィール
A pyrethroid. Tetramethrin is an ester and nitrile. Esters react with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated by the interaction of esters with caustic solutions. Flammable hydrogen is generated by mixing esters with alkali metals and hydrides.
-
农业用途
Insecticide: Tetramethrin is often formulated as an aerosol and
used primarily for indoor pest control or in mosquito coils.
It is also used in shampoos to control fleas and ticks on
pets. It is often formulated with other insecticides and
synergists. Not approved for use in EU countries.
Registered for use in the U.S.
-
製品名
FMC 9260®; ENT-27339; EVERCIDE
INTERMEDIATE® 2265 (tetramethrin + fenvalerate);
MULTICIDE®; NEO-PYNAMIN®; NEOPYNAMINE®;
NEOPYNAMIN FORTE®; NIAGARA®-9260; NIA®-
9260; PHTHALTHRIN®; SP-1103; SUMITOMO®
SP-1103
-
代謝経路
Tetramethrin was the second synthetic pyrethroid to be produced commercially
(1964). The compound has a rapid knock-down action and a
reasonable killing activity which is enhanced by use with synergists. It is
usually used with piperonyl butoxide and in the presence of other insecticides.
It has two chiral centres, both in the acid group, and therefore is a
1 RS-cis-trans mixture. A(1R)-rich product containing cis- and trans-isomers
in the ratio 20:80 is also used in public health; this is named dtetramethrin.
The two products will not be distinguished here because
metabolic studies have been performed with various combinations of
isomers. Most published information relates to its mode of action in
insects and its metabolism in rodents. This is a reflection of its limited
outdoor and field use.