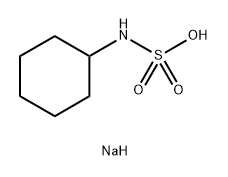
-
外観
白色〜ほとんど白色, 結晶〜結晶性粉末
-
溶解性
水に溶ける。水よく溶け、エタノールに難溶、クロロホルム、ベンゼン、エーテルに不溶。
-
解説
チクロ,サイクラミン酸(シクロヘキシルスルファミン酸)のナトリウム塩,カルシウム塩の通称。合成甘味料の一種。無色〜白色の結晶または結晶性粉末で,水に可溶,砂糖の30〜50倍の甘味をもち,熱に安定。日本では前者は1956年,後者は1961年食品添加物として許可され,広く利用されてきたが,大量摂取は内臓障害や発癌のおそれがあるとされ,1969年使用禁止。その後の追試では発癌性に否定的であった。
-
用途
人工甘味料
-
用途
チクロ(サイクラミン酸ナトリウム)は、人工甘味料のひとつ。IUPAC名はN-シクロヘキシルスルファミン酸ナトリウム sodium N-cyclohexylsulfamate。甘さは砂糖の30倍から50倍といわれる。後味がわずかに苦い(特に高濃度の場合)が、サッカリンやアセスルファムカリウムほどではなく、それらの高甘味度甘味料に比べてすっきりした砂糖に近い甘味をもつ。
-
効能
甘味料 (無栄養素)
-
化学的特性
White powder
-
来歴
Cyclamate was first synthesized in 1937. Like the other sweeteners, its sweet taste was accidentally discovered (U.S. Pat. 2,275,125 (Mar. 3, 1942), L. F. Andrieth and M. Sveda (to E. I. du Pont de Nemours & Co., Inc.). The FDA in 1958 classified sodium cyclamate as a GRAS sweetener. In 1969, a 2-year chronic toxicity study with a sodium cyclamate–sodium saccharin (10:1) mixture found bladder tumors in rats. The FDA took cyclamate off the GRAS list, banning it from foods and beverages, but permitting its sale in pharmacies. In 1970, after a congressional investigation, the FDA banned the use of cyclamate entirely. Abbott Laboratories, which has conducted additional toxicity and carcinogenicity studies with cyclamate, a 10:1 mixture of cyclamate–saccharin, and cyclohexylamine, claimed to be unable to confirm the 1969 findings. Abbott then filed a food additive petition for cyclamate in 1973, which was denied by the FDA in 1980. In 1982, the Calorie Control Council and Abbott Laboratories filed a second food additive petition containing the results of additional safety studies (The Calorie Control Council and Abbott Laboratories, Food Additive Petition for cyclamate 2A3672 (Sept. 22, 1982). That petition remains active.
-
調製方法
Cyclamates are prepared by the sulfonation of cyclohexylamine in
the presence of a base. Commercially, the sulfonation can involve
sulfamic acid, a sulfate salt, or sulfur trioxide. Tertiary bases such as
triethylamine or trimethylamine may be used as the condensing
agent. The amine salts of cyclamate that are produced are converted
to the sodium, calcium, potassium, or magnesium salt by treatment
with the appropriate metal oxide.
-
世界保健機関(WHO)
Cyclamates, non-nutritive sweetening agents, have been used as
additives in food and drugs since 1950. They have been demonstrated to have a
carcinogenic potential at very high and long-sustained dosage in experimental
animals. Some countries have consequently banned their use as food additives,
whereas in others they remain available for this purpose. Most countries, however,
continue to allow their use in small quantities in pharmaceutical preparations.
(Reference: (WHODI) WHO Drug Information, 77.2, 12, 1977)
-
一般的な説明
Odorless or almost odorless white crystals or crystalline powder. Intensely sweet taste, even in dilute solution. pH (10% solution in water): 5.5-7.5. Used as a non-nutritive sweetener.
-
空気と水の反応
Water soluble.
-
反応プロフィール
Sodium N-cyclohexylsulfamate is incompatible with strong oxidizing agents, strong acids and strong bases. Also incompatible with nitrites in acid solution. Has only limited compatibility with potassium salts .
-
危険性
Some evidence of causing cancer in lab-
oratory animals. Prohibited by FDA for food use.
Questionable carcinogen.
-
火災危険
Flash point data for Sodium N-cyclohexylsulfamate are not available; however, Sodium N-cyclohexylsulfamate is probably combustible.
-
応用例(製薬)
Sodium cyclamate is used as an intense sweetening agent in
pharmaceutical formulations, foods, beverages, and table-top
sweeteners. In dilute solution, up to about 0.17% w/v, the
sweetening power is approximately 30 times that of sucrose.
However, at higher concentrations this is reduced and at a
concentration of 0.5% w/v a bitter taste becomes noticeable.
Sodium cyclamate enhances flavor systems and can be used to mask
some unpleasant taste characteristics. In most applications, sodium
cyclamate is used in combination with saccharin, often in a ratio of
10 : 1.
-
安全性プロファイル
Moderately toxic by intravenous and intraperitoneal routes. Mildly toxic by ingestion. Experimental reproductive effects. Questionable carcinogen with experimental neoplastigenic, tumorigenic, and teratogenic data. Human mutation data reported. When heated to decomposition it emits very toxic fumes of NazO, SOx, and NOx.
-
毒性学
Sodium cyclamate is an odorless powder. It is about 30 times as sweet as sucrose in dilute solution. The structure of sodium cyclamate is shown in Figure 10.10 Capillary transitional cell tumors were found in the urinary bladders of 8 out of 80 rats that received 2600 mg/kg body weight per day of a mixture of sodium cyclamate and sodium saccharin (10:1) for up to 105 weeks. When the test mixture was fed at dietary levels designed to furnish 500, 1120, and 2500 mg/ kg body weight to groups of 35 and 45 female rats, the only significant finding was the occurrence of papillar carcinomas in the bladders of 12 of 70 rats fed the maximum dietary level of the mixture (equivalent of about 25 g/kg body weight) for periods ranging from 78 to 105 weeks (except for one earlier death). In vivo conversion from sodium cyclamate to cyclohexylamine was observed particularly in the higher dosage group. Cyclohexylamine is very toxic (LD50 rat oral=157 mg/dg) compared to sodium cyclamate (LD50 oral=12g/kg).
-
貯蔵
Sodium cyclamate is hydrolyzed by sulfuric acid and cyclohexylamine
at a very slow rate that is proportional to the hydrogen ion
concentration. Therefore, for all practical considerations, it can be
regarded as stable. Solutions are also stable to heat, light, and air
over a wide pH range.
Samples of tablets containing sodium cyclamate and saccharin
have shown no loss in sweetening power following storage for up to
20 years.
The bulk material should be stored in a well-closed container in a
cool, dry place.
-
主な応用
Sodium
N-cyclohexylsulfamate (SCHS) dissolved in the aqueous phase
could be introduced as the additive during the interfacial polymerization process. SCHS could not only improve the hydrophilicity and flux of the membrane, but also could enhance the
anti-fouling properties of the membrane[1].
-
規制状況(Regulatory Status)
The use of cyclamates as artificial sweetners in food, soft drinks, and
artificial sweetening tablets was at one time prohibited in the UK
and some other countries owing to concern about the metabolite cyclohexylamine. However, this is no longer the case, and
cyclamates are now permitted for use as a food additive in Europe.
Included in the FDA Inactive Ingredients Database (oral powder,
solutions, chewable tablets, and suspensions). Included in nonparenteral
medicines licensed in the UK. Included in the Canadian List
of Acceptable Non-medicinal Ingredients.