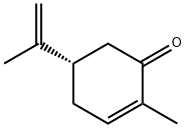
-
用途
香料・香辛料
-
化学的特性
colourless or pale yellow liquid
-
天然物の起源
The optically active and inactive forms have been reported among the constituents of about 70 essential oils.
The dextro form is present in carvi, Antheum graveolens, Antheum sowa, Lippia carviodora, Mentha arvensis, etc. The levo form is
present in Metha vifidis var. crispa, Mentha longifolia from South Africa, Eucalyptus globules and several mint species. The racemic
form is present in ginger grass, Litsea gutalemaleusis, lavender and Artemisia ferganensis. Reported found in citrus oil and juice
(lemon, lime, orange), celery seed, anise, clove, coriander seed, calamus, caraway herb and dill seed.
-
使用
(S)-(+)-Carvone can be used as a starting material to synthesize:
- (-)-Samaderine Y, a pentacyclic quassinoid.
- (-)-Ambrox, a terpenoid responsible for the odor of ambergris.
- 3β-Acetoxydrimenin (a sesquiterpene) via conjugated addition of potassium cyanide followed by base catalyzed Robinson annulation reaction.
- Thapsigargin family members such as trilobolide, nortrilobolide, and thapsivillosin F.
-
定義
ChEBI: A carvone having (S) configuration.
-
一般的な説明
Pale yellow or colorless liquid.
-
空気と水の反応
May be sensitive to prolonged exposure to light and air. Insoluble in water.
-
反応プロフィール
D(+)-Carvone may be sensitive to prolonged exposure to light and air. Incompatible with strong oxidizing agents and strong reducing agents
-
火災危険
D(+)-Carvone is combustible.
-
安全性プロファイル
Poison by ingestion and
skin contact. A skin irritant. When heated to
decomposition it emits acrid smoke and
irritating fumes.
-
職業ばく露
Carvone is found in various natural
oils, including caraway and dillseed; mandarin peel and
spearmint oils. A food additive, it is used in flavoring
liqueurs; in perfumes and soaps.
-
輸送方法
UN2810 Toxic liquids, organic, n.o.s., Hazard
Class: 6.1; Labels: 6.1-Poisonous materials, Technical
Name Required
-
不和合性
Incompatible with oxidizers (chlorates,
nitrates, peroxides, permanganates, perchlorates, chlorine,
bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases,
strong acids, oxoacids, epoxides.