-
解説
リチウムジイソプロピルアミド,C6H14LiN(107.12).略称LDA.ジイソプロピルアミンとブチルリチウムとを低温で反応させて調製し,窒素気流下で用いる.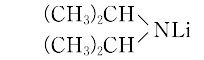 "炭化水素,エーテル系の有機溶媒に可溶.pKa 30以下の酸性水素をもつもの(アセトン,アセトニトリルなど)をリチオ化するのに用いられる.アルキルリチウムよりは求核性が低く,選択性が高い.[CAS 4111-54-0]
"炭化水素,エーテル系の有機溶媒に可溶.pKa 30以下の酸性水素をもつもの(アセトン,アセトニトリルなど)をリチオ化するのに用いられる.アルキルリチウムよりは求核性が低く,選択性が高い.[CAS 4111-54-0]
森北出版「化学辞典(第2版)
-
用途
イソプロピル基のかさ高さのため求核性が低いことを特徴とする。 LDA を反応に用いる常法では、テトラヒドロフラン 7キロバイト
-
化学的特性
Lithium diisopropylamide (LDA) is a white pyrophoric powder. Freshly prepared, it is soluble in hydrocarbons (in hexane about 10 %), but it tends to precipitate irreversibly from solution as a polymer on heating or prolonged storage. In ethers the solubility is much higher, but with the exception of tetrahydropyran, LDA is decomposed at a rate depending on the ether, concentration, and temperature.
Lithium diisopropylamide can be conveniently prepared from butyllithium and diisopropylamine or purchased as a 2 mol/L (25 %) solution in THF and a mixture of various hydrocarbons. Although LDA is unstable in pure THF (at room temperature a 25% solution loses about 1% of its activity per day), the commercially available compositions containing only a limited amount of THF are satisfactory stable for technical applications.
-
使用
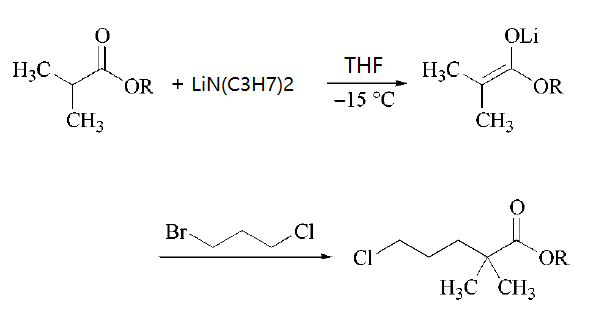
Lithium diisopropylamide is a sterically hindered nonnucleophilic strong base used for selective deprotonations, especially for the production of kinetic enolates (i.e., the thermodynamically less favored isomer) and (hetero-)aromatic carbanions. In the production of the serum lipid regulating agent Gemfibrozil one key intermediate is formed by quenching an ester enolate with 1-bromo-3-chloropropane.
-
一般的な説明
This material is in a solution of THF/Hexanes (ca. 1:7 ratio, respectively)
-
純化方法
It is purified by refluxing over Na wire or NaH for 30minutes and then distilled into a receiver under N2. Because of the low boiling point of the amine, a dispersion of NaH in mineral oil can be used directly in this purification without prior removal of the oil. It is HIGHLY FLAMMABLE, and is decomposed by air and moisture. [Wittig & Hesse Org Synth 50 69 1970, Beilstein 4 H 154, 4 I 369, 4 II 630, 4 III 274, 4 IV 510.]
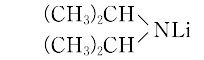 "炭化水素,エーテル系の有機溶媒に可溶.pKa 30以下の酸性水素をもつもの(アセトン,アセトニトリルなど)をリチオ化するのに用いられる.アルキルリチウムよりは求核性が低く,選択性が高い.[CAS 4111-54-0]
"炭化水素,エーテル系の有機溶媒に可溶.pKa 30以下の酸性水素をもつもの(アセトン,アセトニトリルなど)をリチオ化するのに用いられる.アルキルリチウムよりは求核性が低く,選択性が高い.[CAS 4111-54-0]