-
外観
無色~うすい黄色, 澄明の液体
-
溶解性
エタノール及びアセトンに極めて溶けやすく、水にほとんど溶けない。
-
解説
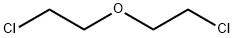
ビス(2-クロロエチル)エーテル,sym-dichloroethyl ether.C4H8Cl2O(143.02).ClCH2CH2OCH2CH2Cl.2,2′-ジクロロエチルエーテルともいう.2-クロロエタノールを濃硫酸で脱水縮合させるか,エチレングリコールに過剰の塩化チオニルを作用させると得られる.クロロホルム様の臭気をもつ無色の油状液体.融点-24.5 ℃,沸点178 ℃.d4201.2199.nD201.4573.引火点79 ℃(開放),55 ℃(密閉).多くの有機溶媒に可溶,水に難溶.ナトリウムエトキシドと反応してジエチレグリコールジエチルエーテルを,第一級アミンと反応してモルホリン誘導体を生じる.毒性があり,緩慢な肺障害を引き起こす.取り扱いには,完全なマスクや保護具の着用が必要である.タール,油脂,ろうなどの溶剤や,土壌の燻(くん)蒸剤として広く用いられる.
-
用途
油脂類溶剤、有機合成中間体、くん蒸剤
-
用途
有機合成中間体、油脂類の溶剤、土壌燻蒸剤(害虫駆除)。
-
化学的特性
colourless liquid
-
物理的性質
Colorless to pale yellow volatile liquid with a pungent, fruity, chlorinated-like odor. The low odor
and high odor threshold concentrations were 90.0 and 2,160 mg/m3, respectively (Ruth, 1986).
-
使用
Reagent for organic synthesis; solvent. Has been used as a scouring agent for textiles; as soil fumigant.
-
一般的な説明
A clear colorless liquid with a sweet pleasant or nauseating odor. Flash point 131°F. Denser than water and insoluble in water. Toxic by inhalation and skin absorption. Used in cleaning compounds, paints, textile finishing, and as a general solvent.
-
空気と水の反応
Flammable. Insoluble in water. Reacts slowly with water to form HCl. Oxidizes readily in air to form unstable peroxides that may explode spontaneously [Bretherick, 1979 p.151-154].
-
反応プロフィール
2,2'-Dichlorodiethyl ether may form phosgene or hydrogen when heated to high temperature. Oxidizes readily in air to form unstable peroxides that may explode spontaneously [Bretherick, 1979 p.151-154]. Mixing in equal molar portions with the following substances in a closed container caused the temperature and pressure to increase: chlorosulfonic acid and oleum [NFPA 1991].
-
健康ハザード
2,2'-Dichlorodiethyl ether is very toxic; the probable oral lethal dose is 50-500 mg/kg, or between 1 teaspoonful and 1 ounce for a 150 pound person. It can be a central nervous system depressant in high concentrations. It is extremely irritating to the eyes, nose, and respiratory passages. It can penetrate the skin to cause serious and even fatal poisoning. Poisonous; may be fatal if inhaled, swallowed or absorbed through skin.
-
火災危険
May form phosgene or hydrogen chloride in fires. There is danger of explosion when ethers are heated or exposed to flames or sparks. Ethers tend to form peroxides; when ethers containing peroxides are heated, they can detonate. May be ignited by heat, sparks, or flames. Container may explode in heat of fire. Vapor explosion and poison hazard indoors, outdoors or in sewers. Decomposes in the presence of moisture to form hydrochloric acid. Emits toxic fumes when heated to decomposition. Reacts vigorously with oleum and chlorosulfonic acid. Ethers tend to form peroxides upon standing. Heating peroxide-containing ethers can cause detonation.
-
化学反応性
Reactivity with Water : No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: V; Inhibitor of Polymerization: Not pertinent.
-
安全性プロファイル
A poison by ingestion,
skin contact, and inhalation. A skin, eye, and
mucous membrane irritant. Questionable
carcinogen with experimental carcinogenic
and tumorigenic data. Mutation data
reported. Exposure to 1000 ppm for 30 to
60 minutes may result in death within days.
The odor is easily detectable at 35 ppm
whch causes only slight irritation.
Flammable liquid when exposed to heat,
flame, or oxidants. Dangerous explosion
hazard; reacts vigorously with oleum,
chlorosulfonic acid. Reacts with water or
steam to evolve toxic and corrosive fumes.
Can react vigorously with oxidizing
materials. To fight fire, use water, foam,
mist, fog, spray, dry chemical. When heated
to decomposition it emits toxic fumes of
Cl-. See also ETHERS.
-
職業ばく露
Dichloroethyl ether is used as a soil
fumigant, an insecticide, and an acaricide; in the manufacture of paint, varnish, lacquer, soap, and finish removers. It
is also used as a solvent for cellulose esters, naphthalenes,
oils, fats, waxes, greases, pectin, tar, and gum; in dry cleaning and in textile scouring.
-
環境運命予測
Biological. When 5 and 10 mg/L of bis(2-chloroethyl)ether were statically incubated
in the dark at 25°C with yeast extract and settled domestic wastewater inoculum, complete
degradation was observed after 7 days (Tabak et al., 1981).
Chemical/Physical. Bis(2-chloroethyl)ether is subject to hydrolysis (Enfield and Yates,
1990; NIOSH, 1994). The hydrolysis rate constant for bis(2-chloroethyl)ether at pH 7 and
25°C was determined to be 2.6 × 10–5/hour, resulting in a half-life o
Emits chlorinated acids when incinerated (Sittig, 1985).
-
輸送方法
UN1916 2,20
-Dichlorodiethyl ether, Hazard
class: 6.1; Labels: 6.1-Poisonous materials, 3-Flammable
liquid
-
純化方法
Wash the ether with conc H2SO4, then Na2CO3 solution, dry with anhydrous Na2CO3, and finally pass it through a 50cm column of activated alumina before distillation. Altern
-
不和合性
Contact with moisture caused decomposition producing hydrochloric acid. Can form peroxides.
May form explosive mixture with air. Contact with strong
oxidizers may cause fire and explosion hazard. Attacks
some plastics, rubber, and coatings. Attacks metals in the
presence of moisture
-
廃棄物の処理
A potential candidate for
liquid injection incineration at a temperature range of 650
to 1600�C and a residence time 0.1 to 2 seconds. Also,
a potential candidate for rotary kiln incineration at
a temperature range of 820 to 1600�C and residence times
of seconds for liquids and gases, and hours for solids. Also,
a potential candidate for fluidized bed incineration at a
temperature range of 450 to 980�C and residence times of
seconds for liquids and gases, and longer for solids.
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste
containing this contaminant (≧100 kg/mo) must conform
with EPA regulations governing storage, transportation,
treatment, and waste disposal