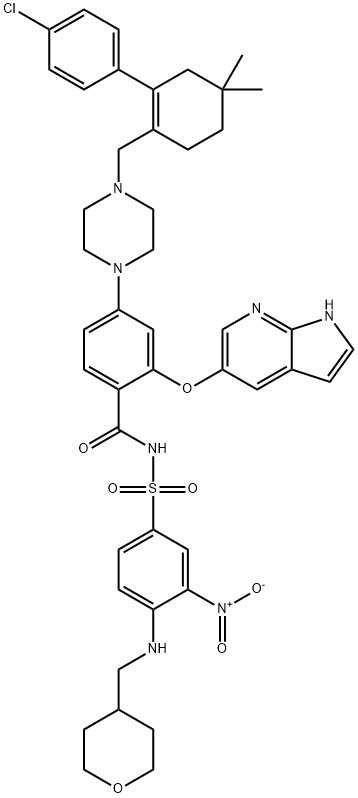
説明
Venetoclax, codeveloped by
AbbVie (previously Abbott Laboratories) and Genentech/
Roche, was approved in the US for treatment of patients with
chronic lymphocytic leukemia (CLL). To meet qualifications
for venetoclax treatment, patients must have received prior
therapy and possess the 17p deletion genetic mutation, as
determined by USFDA testing. Venetoclax functions as a
selective inhibitor of B cell lymphoma subtype 2 (BCL-2),
which is often overexpressed on malignant cells and thus leads
to impairment of the apoptotic pathway. Along these lines,
the orally dosed small molecule drug restores the ability of
malignant cells to undergo apoptosis as its mechanism of
action.90 Although other BCL-2 inhibitors are known, development
of similar agents such as navitoclox have been pursued
and halted due to undesired inhibition of BCL-XL, leading to
significant thrombocytopenia and demonstrating the need for
more selective inhibitors. Venetoclax is also currently being
considered for approval in Europe and Canada for similar
indications and is in various stages of development for the
treatment of non-Hodgkin lymphomas (NHL), acute myeloid
leukemia (AML), multiple myeloma (MM), and several other
disorders, either as a combination therapy or a stand-alone
treatment.