-
効能
抗悪性腫瘍薬, 受容体チロシンキナーゼ阻害薬
-
商品名
ジオトリフ (日本ベーリンガーインゲルハイム)
-
説明
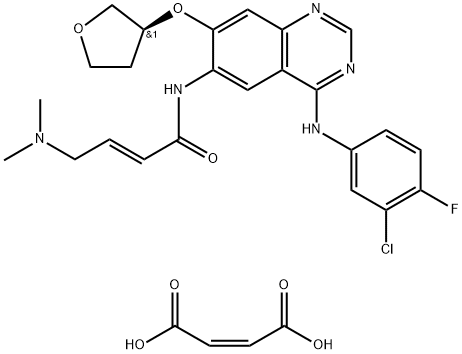
Afatinib dimaleate was approved by the U.S. Food and Drug
Administration (FDA) in 2013 for the treatment of non-small cell
lung cancer (NSCLC). Specifically, it was approved for patients
presenting with metastatic NSCLC tumors which contain epidermal
growth factor receptor (EGFR) exon deletions or exon 21
mutations. Afatinib dimaleate is a covalent inhibitor of ErbB tyrosine
kinases (tyk), which downregulates ErbB signaling by irreversible
binding of EGFR tyk binding sites. While no
manufacturing route has been disclosed to date, the most scalable
published route likely derives from two Boehringer Ingelheim
patents.
-
使用
Afatinib Dimaleate is a salt of Afatinib {BIBW 2992), an aminocrotonylamino-substituted quinazoline derivative used for treating cancer and diseases of the respiratory tract, lungs, gastrointestinal tract, bile duct, and gallbladder. An anilino-quinazoline that irreversibly inhibits EGFR and HER2 kinase activity.
-
定義
ChEBI: Afatinib dimaleate is a maleate salt obtained by combining afatinib with two molar equivalents of maleic acid. Used for the first-line treatment of patients with metastatic non-small cell lung cancer. It has a role as a tyrosine kinase inhibitor and an antineoplastic agent. It contains an afatinib.
-
生物活性
Afatinib dimaleate is an orally bioavailable and irreversible dual-specific inhibitor of the ErbB family (EGFR and HER2). IC50 are 0.4, 0.5, 10, 14, 1 nM for EGFRL858R, EGFRwt, EGFR L858R/T790M, ErbB2 (HER2) and ErbB4 (HER4), respectively. Afatinib dimaleate is available for cancer (esophageal squamous cell carcinoma (ESCC), non-small cell lung cancer (NSCLC) and gastric cancer) studies.