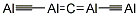-
解説
アルミニウムの炭化物はAl4C3とAl2C6が知られている.三炭化四アルミニウム(tetraaluminium tricarbide):Al4C3(143.96).普通はこちらをさす.Al,またはAl2O3と,炭素粉末を H2 中で電気炉で強熱すると得られる.六方晶系,りょう面体結晶.純粋なら無色であるが,市販品はいくぶん分解して黄色を帯びる.AlC4四面体がつながった複雑な層状構造をしている.密度2.36 g cm-3.モース硬さ8.融点2100 ℃.2200 ℃ 以上で分解する.室温でも希酸に,加熱すると水にも溶けてAl(OH)3とCH4に加水分解する.アルカリ金属,アルカリ土類金属で還元されてAlになる.CH4発生用,金属酸化物の還元剤,触媒,エレクトロニクス基板用のAlNの製造原料などに用いられる.森北出版「化学辞典(第2版)
-
化学的特性
Yellow crystals or powder. Decomposes in water with liberation of methane.
-
使用
Aluminum carbide is used as an abrasive material for cutting tools. It is also used to reduce metal oxides and to prepare aluminum nitride and methane. It and also finds use in metallurgy. Further, it acts as a catalyst and a drying agent.
-
一般的な説明
A yellow powder or crystals. Used to make other chemicals.
-
空気と水の反応
Decomposes slowly in presence of water or moist air to generate heat and flammable methane gas [Merck 12th ed. 1989]. Consequently a dangerous fire risk.
-
反応プロフィール
ALUMINUM CARBIDE is a reducing agent. May react vigorously with oxidizing materials. Incandescence on warming with lead dioxide or potassium permanganate, [Mellor, 1946, Vol. 5, 872].
-
危険性
Dangerous fire risk in contact with moisture.
-
健康ハザード
Inhalation or contact with vapors, substance or decomposition products may cause severe injury or death. May produce corrosive solutions on contact with water. Fire will produce irritating, corrosive and/or toxic gases. Runoff from fire control may cause pollution.
-
火災危険
Produce flammable gases on contact with water. May ignite on contact with water or moist air. Some react vigorously or explosively on contact with water. May be ignited by heat, sparks or flames. May re-ignite after fire is extinguished. Some are transported in highly flammable liquids. Runoff may create fire or explosion hazard.