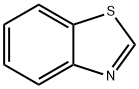
-
外観
無色~赤褐色, 澄明の液体
-
溶解性
水に難溶, エタノール, アセトンに易溶。エタノール及びアセトンに極めて溶けやすく、水にほとんど溶けない。
-
解説
C7H5NS(135.18).ベンゾチアゾールは,N,N-ジメチルアニリンと硫黄とを加熱するか,o-アミノチオフェノールとホルマリンとの反応で得られる.キノリン臭のある液体で,水蒸気蒸留される.沸点227~228 ℃,131 ℃(4.5 kPa).d2041.246.n20D1.6379.エタノール,二硫化炭素に易溶,水に難溶.中性を示す.五塩化リンを作用させると2-クロル誘導体を,混酸でニトロ化すれば6-ニトロ誘導体を与える.森北出版「化学辞典(第2版)
-
用途
有機合成原料。
-
使用上の注意
不活性ガス封入
-
説明
Benzothiazole and its derivative are recognized as the most important heterocyclic compounds. This series of compounds is of particular interests. It has various kinds of pharmacological properties and has been included in many kinds of natural products and pharmaceutical agents. The broad spectrum of pharmacological activity in individual benzothiazole derivative enables it to serve as unique and valuable scaffolds for experimental drug design. It is already known that benzothiazole and its derivative has various biological applications including anticancer, antimicrobial, anticonvulsant, antiviral, antidiabetic, antipsychotic, antiinflammatory, analgesic, fungicidal and diuretic. Its derivatives also have many applications in polymer chemistry, dyes, drugs, and silver photography as well as rubber industry.
-
化学的特性
Benzothiazole has a delicate, persistent, rose-like odor similar to that of quinoline.
-
天然物の起源
Reported found in apricot, cooked asparagus, mozzarella cheese, skim milk powder, roasted beef, beer, malt
whiskey, coconut meat, fresh mango, cooked broccoli and kelp.
-
使用
Various benzothiazole-benzamides synthesized were evaluated for their analgesic and antidepressant activities.
-
定義
ChEBI: An organic heterobicyclic compound that is a fusion product between benzene and thiazole. The parent of the class of benzothiazoles.
-
製造方法
By refluxing a mixture of zinc o-aminophenylsulfide and formic acid, followed by steam distillation of the alkalized reaction
mixture; by heating formanilid or dimethylaniline with sulfur; by oxidation of 2-mercaptobenzothiazole or of the corresponding
disulfide.
-
一般的な説明
Benzothiazole, a volatile sulfur-containing heterocyclic compound, is one of the key odorants of heated milk. It is also reported to occur as a flavor component of miso, coconut meat and cooked-beef.
-
危険性
Highly toxic by ingestion.
-
安全性プロファイル
Poison by ingestion,intraperitoneal, intravenous, and possibly other routes.When heated to decomposition it emits very toxic fumesof SOx, CN??, and NOx.
-
参考文献
Gupta, Akhilesh, and S. Rawat. "Synthesis and cyclization of benzothiazole." J. of Current Pharmaceutical Research 3.1 (2010): 13-25.
Keri, Rangappa S., et al. "A comprehensive review in current developments of benzothiazole-based molecules in medicinal chemistry." European journal of medicinal chemistry 89 (2015): 207-251.
Khokra, Sukhbir L., et al. "Common methods to synthesize benzothiazole derivatives and their medicinal significance: a review." International Journal of Pharmaceutical Sciences and Research 2.6 (2011): 1356.