説明
In December 2012, the US FDA approved bedaquiline as part of combination
therapy for the treatment of multi-drug resistant tuberculosis (MDRTB).
Bedaquiline is the first drug approved for MDR-TB and is the first
approval from a new class of antituberculosis agents in the past 40 years.
Due to the high unmetmedical need for treating MDR-TB, the FDA granted
bedaquiline accelerated approval based on Phase II results, providing patients access to the drug while additional clinical studies are carried out. Bedaquiline (also known as TMC207 and R207910) is a diarylquinoline
that was discovered from a high-throughput, whole-cell screening
strategy with Mycobacterium smegmatis used as a surrogate for
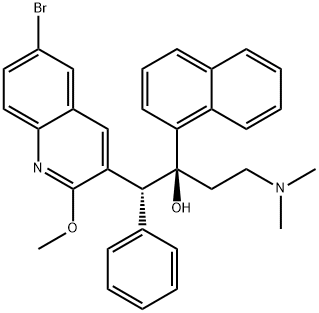
M. tuberculosis. Bedaquiline is a single enantiomer of an initial screening
hit. Bedaquiline has potent and selective activity against mycobacteria, and
is active against both drug-sensitive and drug-resistant M. tuberculosis. The mechanism of action of bedaquiline is unique amongst anti-TB drugs and
involves inhibition of mycobacterial ATP synthase; it is not active against
human ATP synthase. Bedaquiline has in vivo activity in numerous preclinical models of TB infection, alone and in combination with other anti-TB agents, and has bactericidal activity in established TB infection models. Bedaquiline is synthesized in five steps from 3-phenylpropionic acid
and para-bromoaniline. Following amide formation, treatment with POCl3
and DMF under Vilsmeier–Hack conditions gave a 2-chloroquinoline product.
Treatment with sodium methoxide, followed by condensation with
3-(dimethylamino)-1-(naphthalen-1-yl)propan-1-one, and separation of isomers
gave bedaquiline.