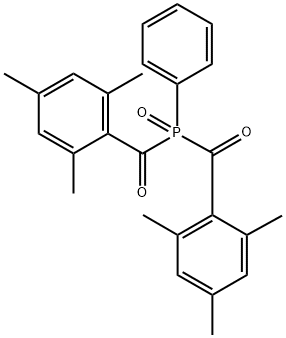
Phenylbis(2,4,6-trimethylbenzoyl) phosphine oxide can be used as a photoinitiator for: Radical polymerization of dental resins. It enhances the polymerization rate and conversion compared to other initiators. It is a polymer-based ceramic material modified with divinylbenzene for the preparation of high-temperature sensor applications.
Phenylbis(2,4,6-trimethylbenzoyl)phosphine oxide is a useful research chemical compound.
Versatile UV photoinitiator for radical polymerization of unsaturated resins, especially pigmented formulations.
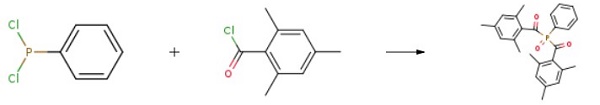
Under nitrogen protection and room temperature, 1.4 g of zinc powder was added to the 100 mL reaction flask with stirring.15 ml of ethyl acetate, 4.1 g of 2,4,6-trimethyl benzoyl chloride, and a solution of 2.5 g of phenylphosphorus dichloride in 5 ml of ethyl acetate was added dropwise to the mixture. After that, the temperature was continued for 2 hours. After the completion of the reaction, 20 mL of water and 1.5 mL of 30% hydrogen peroxide were respectively added to the system, and the reaction was continued for half an hour. Saturated sodium bicarbonate solution was added to neutralize weakly alkaline. Finally, Phenylbis(2,4,6-trimethylbenzoyl)phosphine oxide was obtained after purification.
Phenylbis(2,4,6-trimethylbenzoyl)phosphine oxide (PPO) has a local absorption maximum in the visible spectrum at 405 nm and could provide sufficient initiating radicals to crosslink polyamic diacrylate ester in NMP[1]. Phenylbis(2,4,6-trimethylbenzoyl)phosphine oxide (GR-XBPO) could be used as the reductant, and Monoethanolamine as the oxygen inhibition agent. After UV irradiation, oxygen-containing groups (OCGs) on the graphene oxide (GO) plane and edges are largely removed due to the reduction of GO by free radicals generated by photoinitiator decomposition[2].
[1] Maruti Hegde. “3D Printing All-Aromatic Polyimides using Mask-Projection Stereolithography: Processing the Nonprocessable.” Advanced Materials 29 31 (2017).
[2] Bing Xue, Yuchun Yang, Yingquan Zou. “A UV-light induced photochemical method for graphene oxide reduction.” Journal of Materials Science 52 21 (2017): 12742–12750.