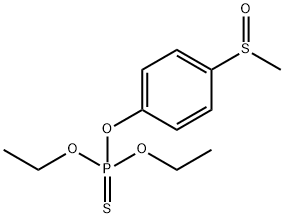
-
外観
ごくうすい黄色~黄色, 澄明の液体
-
溶解性
水に難溶。脂肪族炭化水素以外の殆どの有機溶媒に可溶。脂肪族炭化水素を除く多くの有機溶媒に可溶, 水に難溶(1.54ppm/25℃)※☆。エタノール及びアセトンに溶け、水にほとんど溶けない。
-
用途
農薬(殺虫剤)
-
農薬用途
殺虫剤
-
使用上の注意
アルゴン封入
-
説明
Fensulfothion is a systemic
previously but not currently registered for insecticidal
and nematicidal activity in the United States.
-
化学的特性
Fensulfothion is a yellow oil.
-
使用
Fensulfothion is a highly toxic insecticide used for agriculture purposes.
-
一般的な説明
Oily yellow or brown liquid. Used as an insecticide, nematocide and mosquito larvicide.
-
空気と水の反応
Incompatible with alkali chemicals. Hydrolyzes in alkali, isomerize in air [EPA, 1998]. Slightly soluble in water.
-
反応プロフィール
Organothiophosphates, such as FENSULFOTHION, are susceptible to formation of highly toxic and flammable phosphine gas in the presence of strong reducing agents such as hydrides. Partial oxidation by oxidizing agents may result in the release of toxic phosphorus oxides.
-
危険性
Cholinesterase inhibitor. Questionable carcinogen.
-
健康ハザード
FENSULFOTHION displays cholinesterase inhibiting properties. Death results primarily from respiratory arrest stemming from failure of the respiratory center, paralysis of respiratory muscles and intense bronchoconstriction.
-
火災危険
(Non-Specific -- Organophosphorus Pesticide, Liquid, n.o.s.) FENSULFOTHION may burn but may not ignite readily. Containers may explode in heat of fire. When heated highly toxic fumes of phosphorus and sulfur oxides are emitted. Incompatible with alkali chemicals. Hydrolyzes in alkali, isomerizes in air.
-
安全性プロファイル
A poison by ingestion,
inhalation, and skin contact. Experimental
reproductive effects. A pesticide. When
heated to decomposition it emits very toxic
fumes of SOx and POx.
-
職業ばく露
A potential danger to those involved
in the manufacture, formulation, or application of this
insecticide used to control parasitic, sedentary, and freeliving nematodes.
-
環境運命予測
Soil. In soils, the bacterium Klebsiella pneumoniae degraded fensulfothion to fensulfothion sul?de (Timms and MacRae, 1982, 1983). The following microorganisms were also capable of degrading the parent compound to the corresponding sul?de: Escherichia coli, Pseudomonas ?uorescens, Nocardia opaca, Lactobacillus plantarum and Leuconostoc mesenteroides (Timms and MacRae, 1983).
Plant. Readily oxidized in plants to the corresponding sulfone (Hartley and Kidd, 1987).
Chemical/Physical. Emits toxic fumes of phosphorus and sulfur oxides when heated to decomposition (Sax and Lewis, 1987; Lewis, 1990).
Isomerizes readily to the O,S-diethyl isomer (Worthing and Hance, 1991). The hydrolysis half-lives of fensulfothion in a sterile 1% ethanol/water solution at 25°C and pH values of 4.5, 6.0, 7.0 and 8.0, were 69, 77, 87 and 58 weeks, respectively (Chapman and Cole, 1982).
-
輸送方法
UN3018 Organophosphorus pesticides, liquid,
toxic, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.
UN2810 Toxic liquids, organic, n.o.s., Hazard Class: 6.1;
Labels: 6.1-Poisonous materials, Technical Name Required
-
不和合性
Incompatible with oxidizers (chlorates,
nitrates, peroxides, permanganates, perchlorates, chlorine,
bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases,
strong acids, oxoacids, epoxides. Strong oxidizers may cause
release of toxic phosphorus oxides. Organophosphates, in the
presence of strong reducing agents such as hydrides, may
form highly toxic and flammable phosphine gas. Keep away
from alkaline materials.
-
廃棄物の処理
Alkaline hydrolysis. In
accordance with 40CFR165, follow recommendations for
the disposal of pesticides and pesticide containers. Must be
disposed properly by following package label directions or
by contacting your local or federal environmental control
agency, or by contacting your regional EPA office.