-
外観
白色~うすい灰色又はうすい紫色、粉末
-
性質
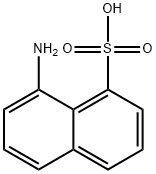
C10H9NO3S(223.25).アミノナフタレン-1-スルホン酸, ペリ酸ともいう.1-ナフタレンスルホン酸をニトロ化した後,還元してアミノ基に変換し,還元液を pH 4.5にすると析出する.母液には副生した5-アミノ誘導体(ローレント酸)が残る.針状晶(一水和物).酢酸に易溶,水,熱湯に難溶.水と200 ℃ に加熱すると1-ナフトール-8-スルホン酸を,水酸化ナトリウム水溶液を220~260 ℃ に加熱すると1,8-ナフタレンジオールを生成する.黒色のアゾ染料,緑色硫化染料の中間原料.森北出版「化学辞典(第2版)
-
溶解性
水に難溶、酢酸に易溶。水酸化ナトリウム溶液に溶ける。
-
用途
黒色アゾ染料、緑色硫化染料の中間体。
-
化学的特性
Dark green powder. 1-Aminonaphthalene-8-sulfonic acid [82- 75-7]. (8-aminonaphthalene-1-sulfonic acid), Peri acid, C10H9NO3S, Mr 223.24, crystallizes as the monohydrate and is sparingly soluble even in boiling water. The sodium salt is also sparingly soluble in water. Caustic fusion at 200℃ produces 8-amino-1- naphthol, whereas slow hydrolysis with 10 % sodium hydroxide at 220 – 260℃ yields 1,8- dihydroxynaphthalene. Sulfonation with cold 10 % oleum produces 1-aminonaphthalene-4,8- disulfonic acid, whereas hot concentrated sulfuric acid results in slow isomerization into naphthionic acid.
-
使用
Intermediate of dyestuff. Dehydration with phosphorus oxychloride yields naphthosultam, and diazotization followed by hydrolysis results in naphthosultone, the intermediate for 1- hydroxynaphthalene-8-sulfonic acid. Diazotization followed by a Sandmeyer cyanide reaction gives 8-cyanonaphthalene-1-sulfonic acid, one of the most industrially important Sandmeyer reaction products because the nitrile forms the lactam, naphthostyril [130-00-7], upon treatment with concentrated alkali at 185℃. Hydrolysis of naphthostyril with dilute alkali yields 8-aminonaphthalene-1-carboxylic acid, which is the key intermediate for production of anthanthrone vat dyes via 1,10 -binaphthyl8,80 -dicarboxylic acid.