-
外観
白色~わずかにうすい黄色, 結晶性粉末~粉末
-
溶解性
エタノール及びメタノールに極めて溶けやすく、水及び酢酸エチルに溶けにくい。
-
用途
プロスタマイド受容体(PM
受容体)に作用し、眼房水の排出を促進する
ことで、眼圧低下作用を示します。
-
効能
抗緑内障薬, 睫毛発毛薬
-
商品名
グラッシュビスタ (アラガン・ジャパン); グラッシュビスタ (アラガン・ジャパン); ルミガン (千寿製薬)
-
説明
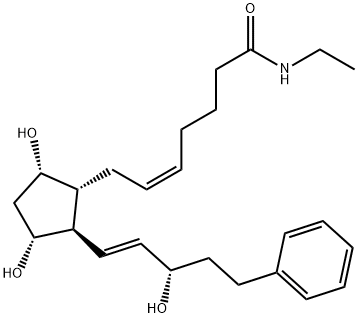
Bimatoprost was first introduced in the US and Brazil as Lumigan?, an ophthalmic
solution (0.03%) for the reduction of elevated intraocular pressure (IOP) in patients with
open-angle glaucoma or ocular hypertension, a proven risk of glaucomatous visual field
loss. Bitamoprost is a new agent belonging to the PGF2α analog class of prostamides
launched in this indication after latanoprost, the most efficaceous topical medication
currently available. This synthetic fatty acid amide can be prepared by esterification of 17-
phenyl-18,19,20-trinorprostaglandin F2α followed by ethylamidation. Bimatoprost, as related
prostamides, is devoid of typical activities associated with PGF2,, analogs; it exhibits a
unique pharmacological profile in contracting the feline iris sphincter with an EC50 of 34 nM
without interacting with any known prostanoid receptors. Thus it mimics the action of
endogenous prostamides on specific receptors that lower IOP by increasing aqueous
humor outflow through both pressure-insensitive and pressure-sensitive pathways without
reducing humor formation. In a 3-month controlled clinical trial of efficacy and safety in patients with elevated IOP, bimatoprost demonstrated lower mean intraocular pressures at
every time point throughout the study, as well as a good tolerance and systemic safety
profile compared to latanoprost.
-
化学的特性
Crystalline Solid
-
使用
Bimatoprost is a prostaglandin analog used topically (as eye drops) to control the progression of glaucoma and in the management of ocular hypertension. It reduces intraocular pressure (IOP) by increasing the outflow of aqueous fluid from the eyes. It ha
-
一般的な説明
Bimatoprost (Lumigan) is supplied as a sterile 0.03%ophthalmic solution in 2.5- and 5.0-mL sizes. The recommendeddosage of bimatoprost is limited to one drop intothe affected eye once daily in the evening. Increased usagemay decrease its beneficial effect. If used concurrently withother IOP-lowering drugs, a waiting period of 5 minutesshould separate administrations.