-
効能
抗悪性腫瘍薬, 受容体チロシンキナーゼ阻害薬
-
商品名
カプレルサ (サノフィ)
-
説明
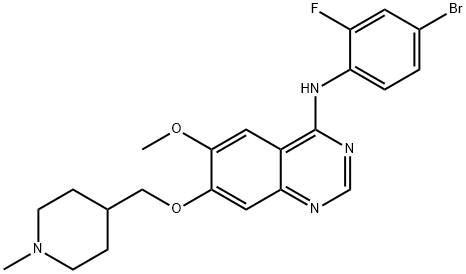
In April 2011, the U.S. FDA approved vandetanib (ZD6474) for the treatment of symptomatic or progressive medullary thyroid cancer (MTC) in adult patients with inoperable advanced ormetastatic disease. Vandetanib inhibits KDR/VEGFR2, VEGFR3, EGFR, and RET kinases with IC50's of 40, 110, 500, and <100 nM, respectively. In athymic mice bearing MTC tumors, a 14.5-fold reduction of tumor volume was observed after 45 days of treatment with vandetanib at 50 mg/kg/day. The decrease in tumor volume was accompanied by decreases in mitotic index (Ki67) and tumor angiogenesis in treated xenografts. Key steps in the synthesis of vandetanib include the displacement of the chlorine atom from 7-benzyloxy-4-chloro-6-methoxyquinazoline with 4-bromo-2- fluoroaniline under acidic conditions in a protic solvent and a Mitsunobu reaction of a N-protected piperidine alcohol with a phenol.
-
化学的特性
Yellow Solid
-
使用
Vandetanib is a once-daily oral inhibitor of vascular endothelial growth factor receptor-2 and epidermal growth factor receptor kinase activity. The activity of Vandetanib plus Docetaxel was assessed
in patients with previously treated non-small-cell lung cancer (NSCLC).
-
臨床応用
Vandetanib, an oral VEGF, EGF, and RET receptor tyrosine kinase
inhibitor, was developed by AstraZeneca for the treatment of
symptomatic or aggressive medullary thyroid cancer (MTC) in patients
with advanced or metastatic disease. This is the first drug
approved for the treatment of MTC. Trials for other cancer indications
such as small-cell lung cancer (SCLC), breast cancer, head and
neck cancer, colorectal cancer, hormone-resistant prostate cancer,
and papillary thyroid cancer are currently being explored. While
AstraZeneca had previously developed ZD-4190 which displays
similar efficacy and pharmacokinetic profile to vandetanib, vandetanib
exhibited significantly improved solubility.