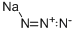-
外観
白色〜ほとんど白色, 結晶性粉末又は塊
-
性質
アジ化ナトリウムは水や液体アンモニアにはよく溶けますが、やエーテルには難溶です。
金属と触れると爆発する危険性があるため、秤量する際はプラスチック製のスパーテルを使用することが推奨されます。
消防法では第5類自己反応性物質として、労働安全衛生法の名称等を表示すべき危険有害物に指定、さらに毒物及び劇物取締法でも毒物に指定されています。
アジ化ナトリウムを吸入または経口ばく露した場合、めまいや呼吸困難、痙攣などの症状を引き起こす恐れがあります。取り扱い場合は、火災や爆発、ばく露に注意することが必要です。
-
反応性
アジ化ナトリウムは熱力学的に不安定な物質であるため、融点を超える温度によってと窒素に分解します。これを利用して、先に述べたように過去にはエアバックで使用されていました。
またアジ化ナトリウムは、酸に反応し有毒で爆発性あるアジ化水素(HN3)を発生させます。
アジ化水素はアジ化ナトリウムと同等の毒性を発揮することに加えて、血管拡張や気管支炎を引き起こす恐れがあるため十分な注意が必要です。
-
溶解性
水に溶けやすく、エタノール及びジエチルエーテルにほとんど溶けない。
-
解説
アジ化ナトリウム.ナトリウムアミドと一酸化二窒素を反応させてつくる.2NaNH2 + N2O → NaN3 + NaOH + NH3無色,六方晶系の結晶.密度1.846 g cm-3.水に易溶.打撃では爆発しないが,加熱すると分解し,Naと N2 になるので純粋なナトリウムの原料になる.酸を加えるとアジ化水素を生じる.シトクロム・オキシダーゼの抑制作用がある.長く接触すると皮膚や眼にかゆみを生じ,摂取すると,おう吐・呼吸困難・頭痛・心悸亢進などを起こす.ほかのアジ化物の原料のほか,医薬品,農薬,防腐剤,分析試薬,自動車のエアバッグや航空機の緊急脱出シュートを膨らませるための起爆剤などに用いられる.有毒.
-
用途
還元剤、発色試薬、金属アジ化物製造原料、調製液原料(安定剤、防腐剤)。有機合成(和光試薬時報Vol.66 No.2,p.21(1998)、Vol.65 No.4,p.30(1997))。
-
用途
診断薬、爆発原料、有機合成原料、自動車用エアーバッグの推進薬
-
製法
工業的には、Wislicenus process と呼ばれる方法で製造されています。これは、液体アンモニウムから(NaNH2)を経て (N2O) と反応させる製造法です。
参考文献
-
使用上の注意
酸性溶液を加熱すると有害なアジ化水素を発生する。
-
化学的特性
Sodium azide is a colorless to white, odorless, crystalline solid. Combustible solid above 300°C.
-
物理的性質
Colorless hexagonal crystals; density 1.846 g/cm3 at 20°C; decomposes on heating to produce sodium and nitrogen; also decomposes in vacuum; soluble in water partially converting to hydrazoic acid, solubility in water, 41.7 g/100mL; slightly soluble in alcohol, 0.316g/100mL at 16°C; soluble in liquid ammonia.
-
使用
Sodium azide is used in making othermetal azides, therapeutically to control bloodpressure, as a propellant for automotivesafety bags, as a preservative for laboratoryreagents, as an analytical reagent, andin organic synthesis. It is also used as anantifading reagent for immunofluorescence(Boeck et al. 1985).
-
定義
sodium azide: A white or colourlesscrystalline solid, NaN3, soluble inwater and slightly soluble in alcohol;hexagonal; r.d. 1.846; decomposes onheating. It is made by the action ofnitrogen(I) oxide on hot sodamide(NaNH2) and is used as an organicreagent and in the manufacture ofdetonators.
-
調製方法
Sodium azide can be prepared from sodium metal and liquid
ammonia in the presence of ferric chloride. The amide
formed is treated with nitrous oxide to produce the azide.
-
製造方法
Sodium azide is prepared by reacting sodium amide with nitrous oxide. The amide is heated with nitrous oxide at 200°C or its solution in liquid ammonia is treated with nitrous oxide at ambient temperature: 2NaNH2 + N2O → NaN3 + NaOH + NH3.
-
反応プロフィール
Sodium azide is unstable. Decomposes rapidly or explosively at about 300°C [Hawley]. May explode if shocked. Forms violently explosive products if exposed to carbon disulfide. Can be sensitized toward decomposition by metal salts (especially heavy metal salts such as silver chloride) or by traces of strong acids [Sax, 9th ed., 1996, p. 298].
-
危険性
Sodium azide is a toxic as well as an explosive substance (Patnaik, P. 1999. A Comprehensive Guide to the Hazardous Properties of Chemical Substances, 2np ed. New York: John Wiley & Sons). Although inert to shock, violent decomposition can occur when heated at 275°C. Contact of solid or solution with lead and copper must be avoided. Reactions with halogens, carbon disulfide, or chromyl chloride can be explosive. Dissolution in water produces toxic vapors of hydrazoic acid. The salt is an acute poison causing headache, hypotension, hypothermia, and convulsion. LD50 oral (rats): 27 mg/kg.
-
健康ハザード
The acute toxicity of sodium azide is high. Symptoms of exposure include lowered blood pressure, headache, hypothermia, and in the case of serious overexposure, convulsions and death. Ingestion of 100 to 200 mg in humans may result in headache, respiratory distress, and diarrhea. Target organs are primarily the central nervous system and brain. Sodium azide rapidly hydrolyzes in water to form hydrazoic acid, a highly toxic gas that can escape from solution, presenting a serious inhalation hazard. Symptoms of acute exposure to hydrazoic acid include eye irritation, headache, dramatic decrease in blood pressure, weakness, pulmonary edema, and collapse. Solutions of sodium azide can be absorbed through the skin. Sodium azide has not been found to be carcinogenic in humans. Chronic, low-level exposure may cause nose irritation, episodes of falling blood pressure, dizziness, and bronchitis.
-
火災危険
When heated to decomposition, Sodium azide emits very toxic fumes of nitrogen oxides; explosive. Forms explosive-sensitive materials with some metals such as lead, silver, mercury or copper. May form toxic hydrazoic acid fumes in fire. Containers may explode in fire. Avoid acids, benzoyl chloride and potassium hydroxide; bromine; carbon disulfide; copper; lead; nitric acid; barium carbonate; sulfuric acid; chromium (II) hypochlorite, dimethyl sulfate, water, dibromomalononitrile, lead, silver, copper, mercury. Hazardous polymerization may not occur.
-
燃焼性と爆発性
Flammability hazard is low, but violent decomposition can occur when heated to
275 °C. Decomposition products include oxides of nitrogen and sodium oxide.
-
使用用途
アジ化ナトリウムの主な使用用途には以下の5つがあります。
- アジド基の導入
アジ化ナトリウムはアジ化物イオンとナトリウムイオンで構成される塩です。そのため、アジ化物イオンの求核性を利用することで、有機化合物にアジド基を導入することができます。
- 窒素ガス発生剤
アジ化ナトリウムは爆発原料としても使用されています。以前までは、自動車の運転席側エアバックにおいて、衝突時などの衝撃で火薬を爆発させることで生じる高熱を利用して、アジ化ナトリウムを爆発させて窒素ガスを発生させる発生剤に使用されていました。
- 防腐剤
アジ化ナトリウムは市販の抗体に防腐剤として含まれていることがあります。これによって細菌の増殖を抑えることが可能になります。しかし、アジ化ナトリウムはホースラディッシュ・ペルオキシダーゼ (Horseradish peroxidase; HRP) という酵素の活性を阻害することから、HRPの標識抗体には含まれていません。
- 溶存酸素 (DO) の測定
水中の溶存酸素量 (DO) を測定する方法の1つに「ウィンクラー法」があります。アジ化ナトリウムを試料中に加えておくことで、亜硝酸イオンとDOの反応を抑制することが出来ます。
-
安全性プロファイル
Poison by ingestion, skin contact, intraperitoneal, intravenous, and subcutaneous routes. Human systemic effects by ingestion: general anesthesia, somnolence, and hdney changes. Questionable carcinogen with experimental tumorigenic data. Human mutation data reported. Violent reaction with benzoyl chloride combined with KOH, Br2, barium carbonate, CS2, Cr(OCl)2, Cu, Pb, HNO3, BaCO3, H2SO4, hot water, (CH3)2SO4, dibromomalononitrile, sulfuric acid. Incompatible with acids, ammonium chloride + trichloroacetonitrile, phosgene, cyanuric chloride ,2,5 -dinitro3 methylbenzoic acid + oleum, trifluroroacryloyl chloride. Reacts with heavy metals (e.g., brass, copper, lead) to form dangerously explosive heavy metal azides, a particular problem in laboratory equipment and drain traps. When heated to decomposition it emits very toxic fumes of NOx and Na2O. See also AZIDES.
-
職業ばく露
Sodium azide is used as preservative and diluent. It has been used for a wide variety of military, laboratory, medicine, and commercial purposes. It is used extensively as an intermediate in the production of lead azide, commonly used in detonators, and other explosives. Reported to be used in automobile air-bag inflation. One of the largest potential exposure is that to automotive workers, repairmen, and wreckers, if sodium azide is used as the inflation chemical. Commercial applications include use as a fungicide, nematocide, and soil sterilizing agent and as a preservative for seeds and wine. The lumber industry has used sodium azide to limit the growth of enzymes responsible for formation of brown stain on sugar pine, while the Japanese beer industry used it to prevent the growth of a fungus which darkens its product. The chemical industry has used sodium azide as a retarder in the manufacture of sponge rubber, to prevent coagulation of styrene and butadiene latexes stored in contact with metals; and to decompose nitrites in the presence of nitrates.
-
貯蔵
In particular,
work with sodium azide should be conducted in a fume hood to prevent exposure by
inhalation, and appropriate impermeable gloves and splash goggles should be worn
at all times to prevent skin and eye contact. Containers of sodium azide should be
stored in secondary containers in a cool, dry place separated from acids.
-
輸送方法
UN1687 Sodium azide, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.
-
純化方法
Crystallise sodium azide from hot water or from water by adding absolute EtOH or acetone. Also purify it by repeated crystallisation from an aqueous solution saturated at 90o by cooling it to 10o, and adding an equal volume of EtOH. The crystals are washed with acetone, and the azide is dried at room temperature under vacuum for several hours in an Abderhalden pistol. Its solubility in H2O is 42% at 18o, and in EtOH it is 0.22% at 0o. [Das et al. J Chem Soc, Faraday Trans 1 78 3485 1982, Schenk in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I pp 474-475 1963, Browne Inorg Synth 1 79 1939, Frierson Inorg Synth II 139 1946.] HIGHLY POISONOUS and potentially explosive.
-
不和合性
Reacts explosively and/or forms explosive and/or shock sensitive compounds with acids and many metals. Contact with water forms hydrazoic acid. Combustible solid (if heated above 275�C). May explode when heated above its melting point, especially if heating is rapid. Reacts with acids; producing toxic, shock-sensitive, and explosive hydrogen azide. It forms explosive compounds with phosgene, brass, zinc, trifluoroacrylol fluoride, and nitrogendiluted bromine vapor. Reacts with benzoyl chloride and potassium hydroxide, bromine, carbon disulfide; copper, lead, nitric acid; barium carbonate; sulfuric acid; chromium (II) hypochlorite; dimethyl sulfate; dibromomalononitrile, silver, mercury. Over a period of time, sodium azide may react with copper, lead, brass, or solder in plumbing systems to form an accumulation of the highly explosive and shock-sensitive compounds of lead azide and copper azide
-
廃棄物の処理
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform to EPA regulations governing storage, transportation, treatment, and waste disposal. Disposal may be accomplished by reaction with sulfuric acid solution and sodium nitrate in a hard rubber vessel.Nitrogen dioxide is generated by this reaction and the gas is run through a scrubber before it is released to the atmosphere. Controlled incineration is also acceptable (after mixing with other combustible wastes) with adequate scrubbing and ash disposal facilities.