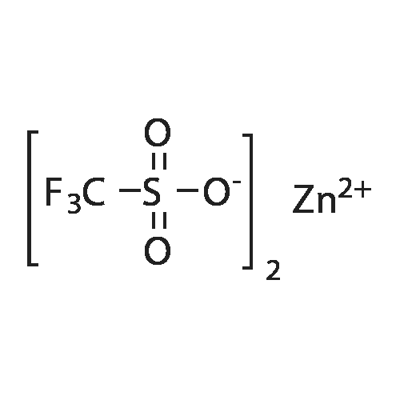
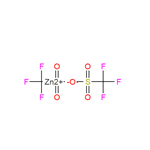
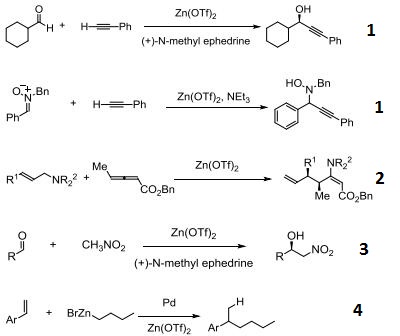
Reaction
- Catalyst for the addition of acetylenes to carbonyls and nitrones.
- Claisen rearrangement.
- Catalyst for the enantioselective Henry and Aza-Henry reactions.
- Pd-catalyzed hydroalkylation of styrenes with zinc reagents.
Chemical Properties
white to light-grey powder
Uses
Zinc trifluoromethanesulfonate acts as a catalyst for the preparation of dithioketals. It is used as a Lewis acid catalyst in silylation reactions. It is also used as a catalyst for greener amine synthesis by reductive amination with hydrogen gas.
Synthesis
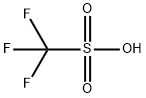
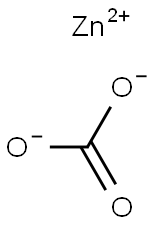
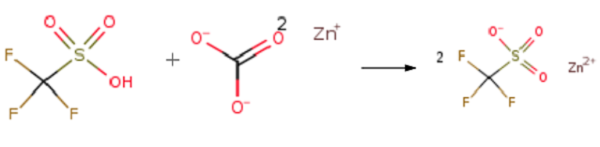
Triflic acid (0.056mol) was added dropwise to a suspension of zinc carbonate (0.02 mol)in dry methanol (20 ml) at room temperature. During the addition, CO2 was evolved. The mixture was stirred at 25°C for 20 minutes and refluxed for 2 h. The clear solution was cooled to 25°C and concentrated under reduced pressure. The resulting white powder was dried at 125°C for 2 h to afford Zinc trifluoromethanesulfonate (Zn(OTf)2 )(98% yield).
Purification Methods
It should be dried at 125o for 2hours at 3mm before use. It is soluble in CH2Cl2 but insoluble in pet ether. [Corey & Shimoji Tetrahedron Lett 24 169 1983.]