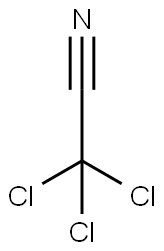
General Description
Clear pale yellow liquid.
Reactivity Profile
May be sensitive to light and heat. This compound may react with water, steam, acid or acid fumes. TRICHLOROACETONITRILE(545-06-2) may hydrolyze under acidic or alkaline conditions. . The reaction of benzene and TRICHLOROACETONITRILE(545-06-2) evolves toxic chloroform and HCl gasses. (Hagedorn, F., H.-P. Gelbke, and Federal Republic of Germany. 2002. Nitriles. In Ullman Encyclopedia of Industrial Chemistry. Wiley-VCH Verlag GmbH & Co. KGaA.).
Air & Water Reactions
Highly flammable. Insoluble in water.
Fire Hazard
This chemical is combustible.
Chemical Properties
colourless to slightly yellow liquid
Uses
Trichloroacetonitrile is involved as a reagent in Overman rearrangement, which is used to prepare alylic amines from allylic alcohols. It is also used to prepare bistrichloroacetimidates from diols leading to dihyrooxazines through acid catalyzed cyclization. Further, it is utilized in the synthesis of trichloroacetimidates by 1,8-Diazobicyclo[5.4.0]undec-7-ene (DBU) catalyzed addition of allylic alcohols. It finds application in the study of the methoxy methyl (MOM) catalyzed aza-Claisen rearrangement.
Definition
ChEBI: Trichloroacetonitrile is an aliphatic nitrile and an organochlorine compound.
Production Methods
Trichloroacetonitrile can be obtained by dehydration of trichloroacetamide with phosphorous pentoxide or by chlorination of acetonitrile with chlorine. Vapor phase chlorination in the presence of water and photochemical chlorination in the presence of catalysts such as HgCl2 or AlCl3 have been reported. Trichloroacetonitrile is an organic intermediate used, for example, in the synthesis of the fungicide etridiazole.
Hazard
Strong irritant to tissue. Questionable carcinogen.